Dynamic search, filtering, and navigation
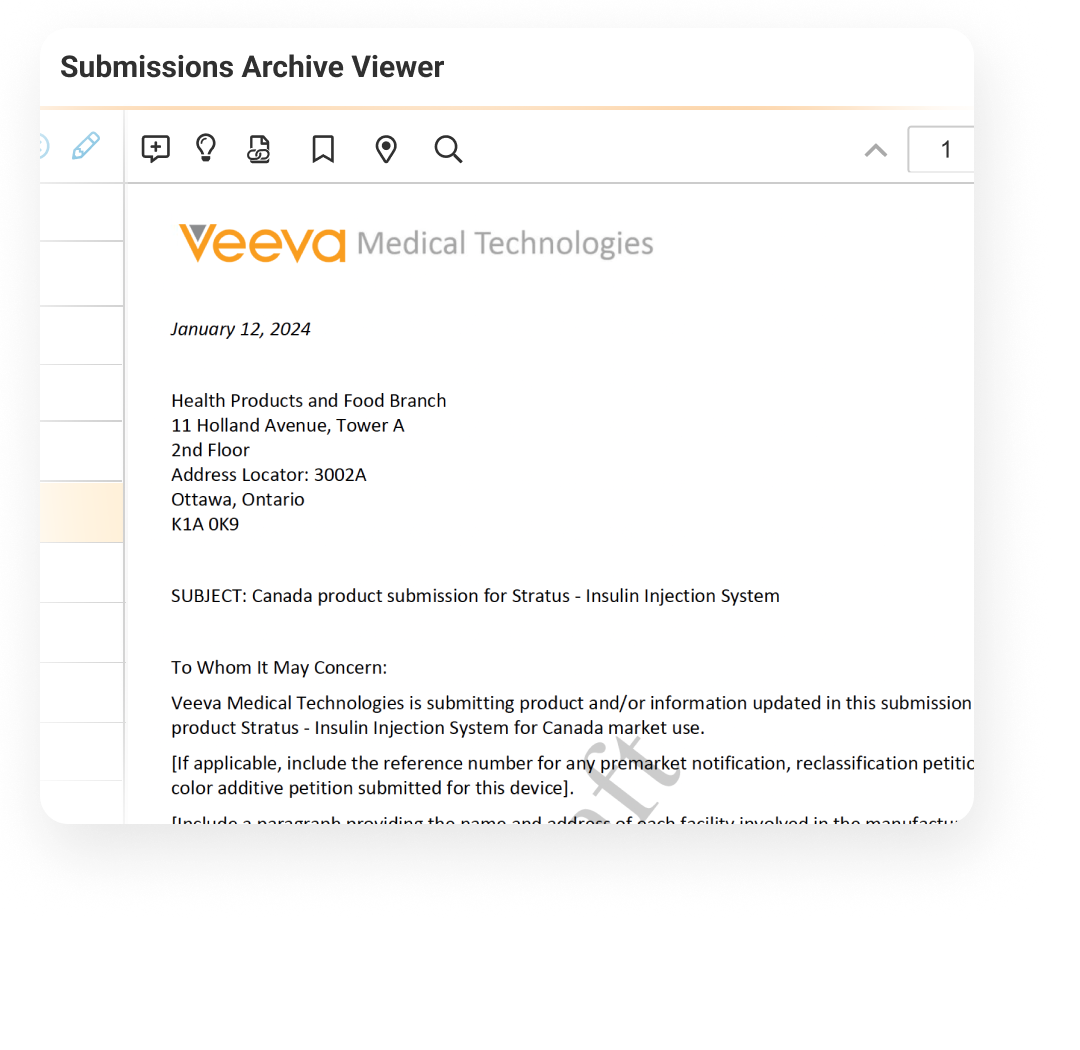
Veeva Submissions Archive is a global, secure repository of submission published output. It functions as the authoritative source of applications submitted to health authorities.
Submissions Archive includes a viewer that supports all electronic and paper formats, with PDF link navigation provided for electronic formats. Users can view submissions and health authority correspondence alongside all previously submitted applications. The embedded document viewer provides visibility into each document in the structure.
The Active Dossier feature displays the submission components that are currently active for any product / market combination.
80%
submission tasks eliminated or simplified
200+
hours saved on approval and renewal reporting
65+
legacy systems consolidated
Veeva Submissions Archive Impact
Control user access
Share the right content with the right people based on configured business rules.Maintain global readiness
Allow remote team members to access authorized submissions from anywhere in the world.Track health authority correspondence and commitments
Easily locate regulatory dossiers including those sent by regional offices or local affiliates to health authorities.
See Veeva Submissions Archive in action
Customer Success
Medtechs ensure compliance with Veeva RIM






Read white paper
See how six leading GenAI models stack up against human experts on typical regulatory affairs tasks

Read buyer's guide
Determine scope, evaluate software capabilities, and balance cost with value for your next RIM project

Read article
Bio-Rad, Thermo Fisher Scientific, and the FDA share how global harmonization efforts benefit the medtech industry

Learn more
Veeva Submissions Archive Features Brief


