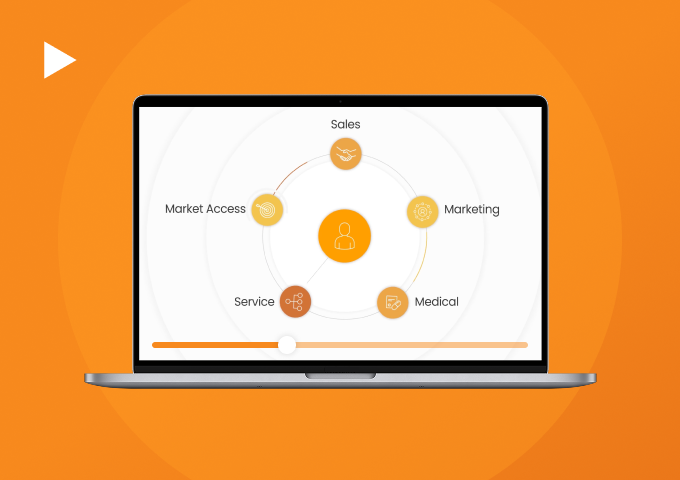
Connect Sales, Marketing, and Medical for Greater Impact
Veeva Commercial Cloud serves as the technology foundation for commercial execution connecting sales, marketing, and medical teams for customer centricity. Leveraging integrated data and Veeva AI, it delivers a comprehensive suite of solutions that accelerate insights, foster innovation, and maximize efficiency.
Veeva Commercial Cloud unifies stakeholder data, commercial and medical content, and customer engagement activity in a single platform while maintaining boundaries required for compliance.

-
Drive End-to-End Content Management
Purpose-built solution for commercial content, orchestrating the entire lifecycle from creation and review to compliant distribution.
-
Achieve Customer Centricity
Coordinate across teams with shared content, data, and engagement channels to create the best experience for customers.
-
Lead with AI-powered Insights
Contextual AI Agents and high-quality data deliver deeper insights, drive faster decision making, and enable personalized interactions.
Products
Veeva Commercial Cloud
Streamline end-to-end content and claims management and enable real-time collaboration to deliver more customer-centric and coordinated engagement.

Veeva Vault CRM
Connect sales, marketing, and medical teams on a single platform
Veeva PromoMats
Speed the creation, review, and distribution of compliant content
Veeva AI
Drive greater efficiency and effectiveness with agentic AI in the Vault Platform and industry-specific application agents
Veeva Link Key People
Enable more relevant engagement with deep KOL data
Veeva OpenData
Power commercial agility with high-quality reference data
75%
Faster content review
2X
the content review capacity
72%
new KOLs found
Watch Video
Establishing the Foundation for MedTech Commercial Excellence
Read eBook
The MedTech CRM Decision: Key Advantages of a Purpose-Built Solution

Read article
Shaping the future of the medtech content supply chain with AI

Customer story
Fresenius Medical Care reduces commercial content approval by 14 days

Read article
Impact of AI in medtech, from real-world evidence to content generation


