Boston Scientific and Philips: How to Ensure First-Time-Right Design by Integrating Quality and Engineering
Experts from Philips and Boston Scientific share strategies for shifting culture, connecting systems, and driving continuous improvement
Medtech companies must move at a rapid pace to meet patient needs without compromising on their commitment to quality. The continuous pressure on R&D to innovate faster, combined with rising regulatory scrutiny, can create product quality issues or potential compliance risks that often manifest as organizational pain. Traditional silos turn quality teams into “police personnel” or “gatekeepers,” causing delays, while engineering teams face crippling rework cycles due to fragmented data. To overcome this costly cycle, quality must evolve from a late-stage gate to an integrated discipline across the product lifecycle.
Robby van Dreumel, director of design quality at Philips, and Olivia Sheahan, senior manager of business transformation at Boston Scientific, highlighted their strategies for achieving this continuous integration.
From gatekeeping to design quality engineering
The foundational shift begins with a strong quality culture that demands shared values and never compromises patient safety. For Van Dreumel, this meant transforming the traditional quality assurance role into design quality engineering (DQE) and moving to a proactive model that focuses on first time-right design.
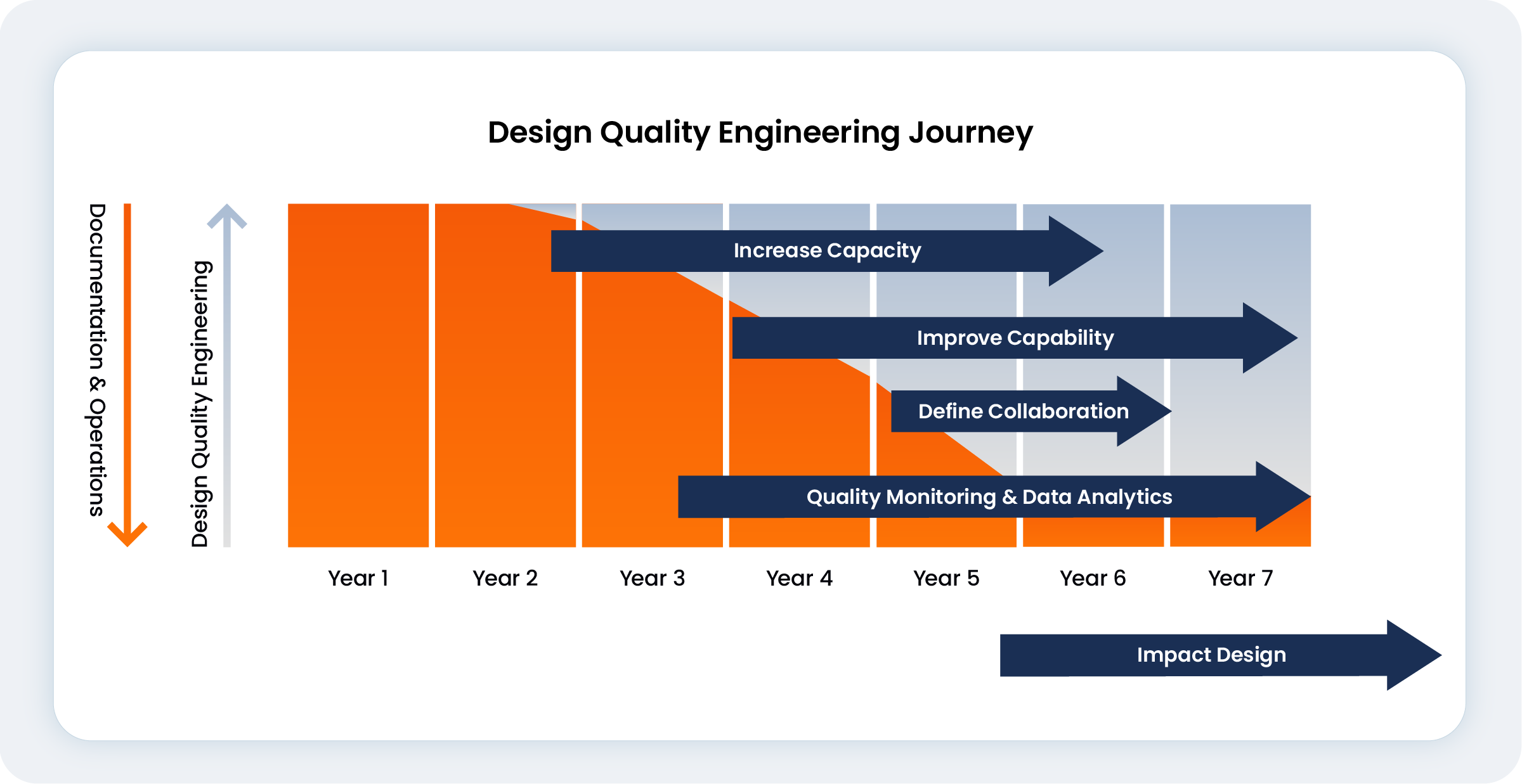
The DQE role moves quality farther upstream by positioning quality as influential partners in development, actively engaging between project milestones. To ensure success in this cross-functional partnership, Van Dreumel saw the potential to grow his DQE team and further train them on systems engineering and crucial soft skills, such as influencing and negotiation. He illustrated this partnership, “I stayed with them, actually helped with the testing, helped with writing the reports… that was a moment where people started to see me as a partner.”
Shared ownership is critical, as Sheahan also notes: “Everyone needs to fundamentally understand that we own it all… it’s teamwork with all our stakeholders. It’s quality engineering, supplier quality, product builders, it’s everybody.”
A foundation of connected systems and data
Specialized systems like PLM (as designed) and QMS (as improved) often silo critical data. Van Dreumel and Sheahan advocate for breaking down these silos to provide quality with real-time visibility and engineering with uninterrupted workflows—a persistent challenge across the industry. Establishing a closed-loop process across these systems will help organizations maintain product integrity from design through production.
Van Dreumel utilized critical to safety (CTS) and critical to quality (CTQ) flowdown to link design, risk, and controls. He noted that this approach provides true end-to-end traceability and connects critical steps across the value chain.
Integrating quality within the supply chain
The final product’s quality depends on its components. Fragmented data triggers painful rework spirals and supplier selections that do not meet quality requirements. As Sheahan observed, the challenge is when teams “end up in these panic modes of rework because we have all the information in front of us and then decisions are made and lock us into decisions that might not meet the business goals.”
Boston Scientific manages supplier quality through a three-legged stool approach: global sourcing, supplier quality, and supplier engineering. To address the core tension of visibility into a supplier’s true quality risk, the company sees value in an integrated solution. The aim is to empower design teams to evaluate comprehensive risk factors—such as previous audits, SCARs, and postmarket data—well before procurement begins. As Sheahan explained, the goal is to ensure “teams are making the right selection… doing it right the first time and we’re not entering into those rework cycles”.
Closing the loop with quality monitoring and analytics
Although an enormous amount of data was available, Van Dreumel highlighted that simply having it was not enough to make an impact. He noted, “There was an opportunity to have insights drive value in the design process by ensuring that quality monitoring and data analytics feed directly back into engineering.”
Van Dreumel took this opportunity to implement a dashboard, linking service replacement rates (QMS data) to the product’s architecture (PLM data). This provides R&D with performance visibility and unexpected insights to act on. Van Dreumel further shared “This deeper focus on our data helped us discover unexpected opportunities that were living elsewhere in our systems.”
Leveraging postmarket data is essential for continuously updating FMEAs and measuring the effectiveness of continuous improvements.
Strategic value of integrated quality
By integrating quality and engineering, Medtech companies can drive patient safety and efficiency while also managing to:
- Reduce rework: Embed quality processes early in the design phase to identify potential failures before they require costly, late-stage corrections.
- Optimize supplier partnerships: Empower teams to manage suppliers based on real-time data, rather than relying on cost or legacy relationships.
- Accelerate speed-to-market: Capture compliance data concurrently with design to ensure faster, more reliable global market access.
For more insights, hear how Teleflex is establishing data-driven quality for patient safety with a unified platform.