Modern EDC for complex trials
Veeva Electronic Data Capture (EDC) provides an end-to-end environment to collect, review, and process trial data about patients and samples.
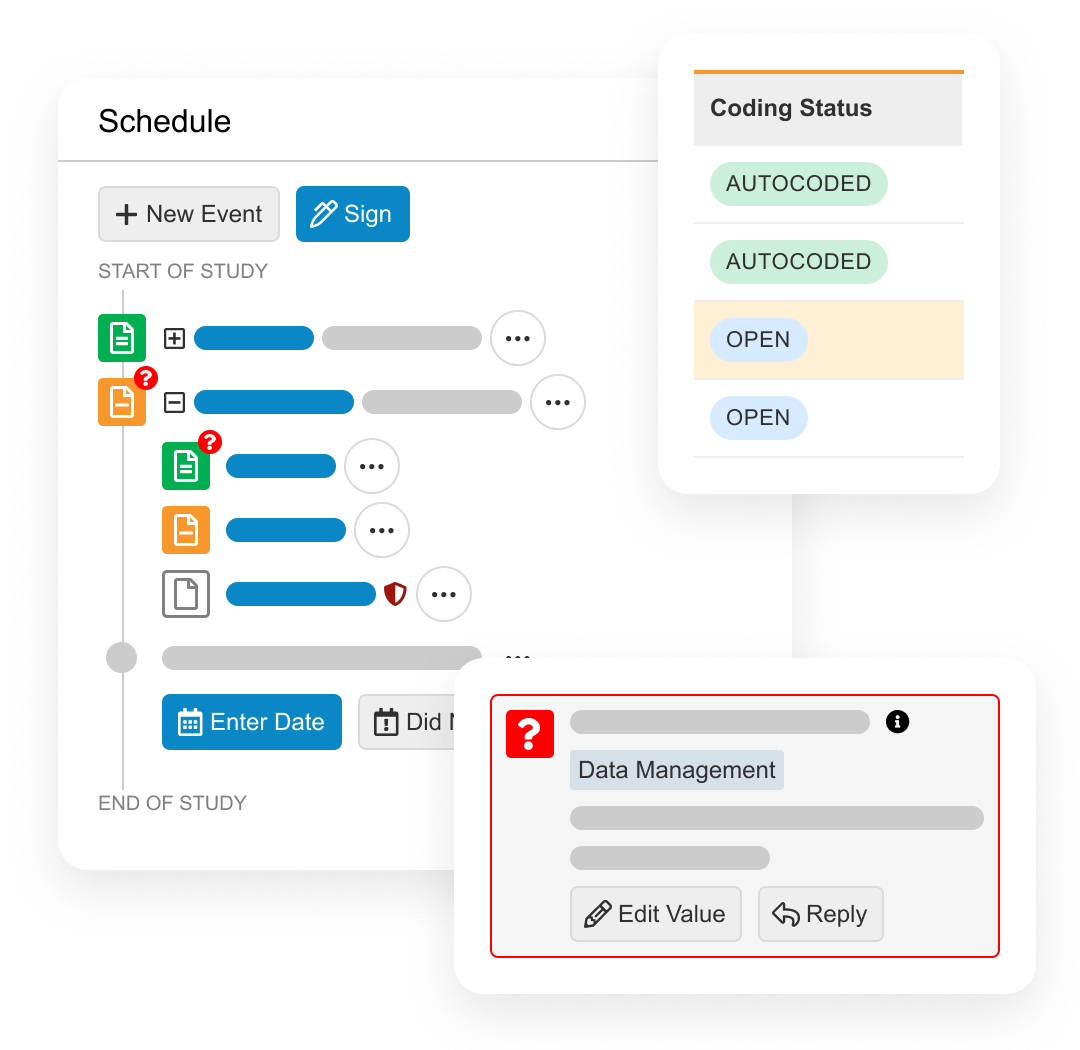
During study start, Veeva EDC is used to design case report forms (including edit checks) without the need for custom programming.
During study execution, Veeva EDC collects all patient form data, local labs, DICOM images, and medical coding. It also includes quality controls for querying, targeted source data verification (SDV), and protocol deviations. When protocol amendments happen, the Veeva EDC database needs no downtime.
At the end of the study, Veeva EDC provides data lock and post-processing features, including end-of-study media creation and archiving.
50%
faster study builds
100%
elimination of known custom functions
9x
faster to implement study changes
Veeva EDC Impact
Build studies faster
Design complex studies with an in-product specification studio built on Agile Design principles.Get cleaner data with less effort
Speed SDV and medical assessments with role-based user interfaces that prompt action.Streamline image management
Securely store, manage, and retrieve images with a streamlined solution for DICOM image management.Run complex trials with no downtime
Flexibly design and operate all studies, including master protocols and adaptive designs.
See Veeva EDC in action
Customer Success
Medtechs manage complex trials with Veeva Clinical Data
































Read article
Alcon and Illumina share insights on the challenges, best practices, and outcomes of implementing Veeva EDC

Watch video
Cochlear shares how they drive operational excellence and create a set of key metrics with Veeva's unified platform

Read customer story
Global medical device company transforms how clinical trials are built and managed with Veeva EDC

View infographic
The Clinical Benchmark studies how organizations manage clinical processes, study site collaboration, and trial data

Read white paper
Learn how organizations transform database design with a modern EDC

Read customer story
Learn how the Veeva Clinical Platform transforms how clinical trials are built and managed







