Why Unified Data is the New Standard in Medtech Medical Affairs
The shift from fragmented, siloed systems to a unified deep data foundation is now the industry standard for reducing financial risk and accelerating the delivery of medical innovations to patients. By establishing a single source of truth, organizations can eliminate operational friction, enhance cross-department alignment, and ensure AI readiness for a more competitive, data-driven future.
In medtech, the product development lifecycle depends on the quality of insights surrounding Key Opinion Leaders (KOLs). From initial design to market adoption, a connected view of an expert’s scientific activity and professional network is a critical differentiator—especially as AI enters the mix. In this complex landscape, unreliable data is more than an administrative headache; it is a financial risk and a potential delay in getting innovations to patients.
Despite what is at stake, many medtech medical leaders operate within a fragmented digital landscape. Teams often rely on siloed platforms for data collection and sharing, creating friction where there should be flow. Recent data highlights this disconnect, showing that 59% of organizations are still navigating a landscape where tools are either independent or limited to basic infrastructure.
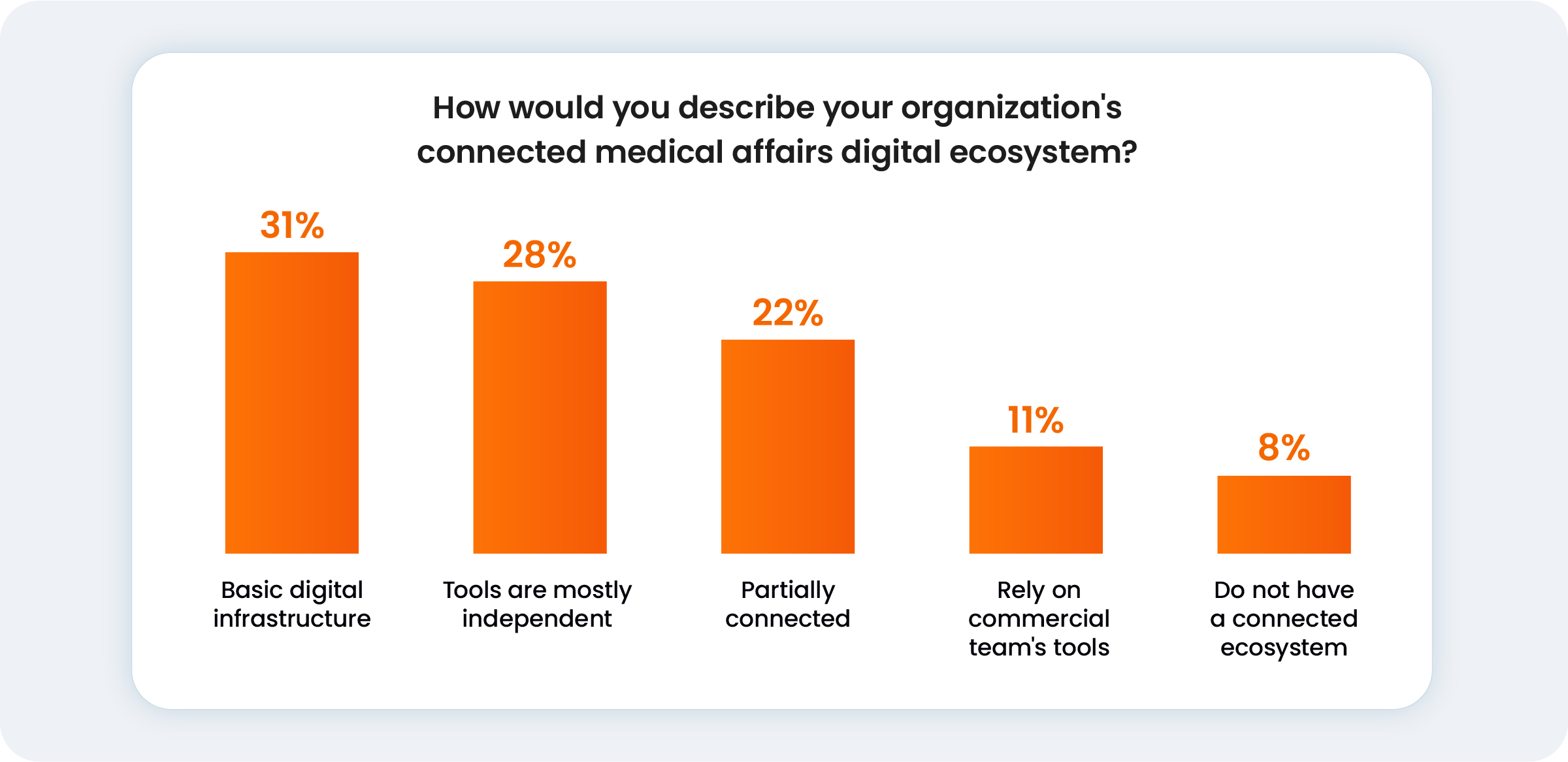
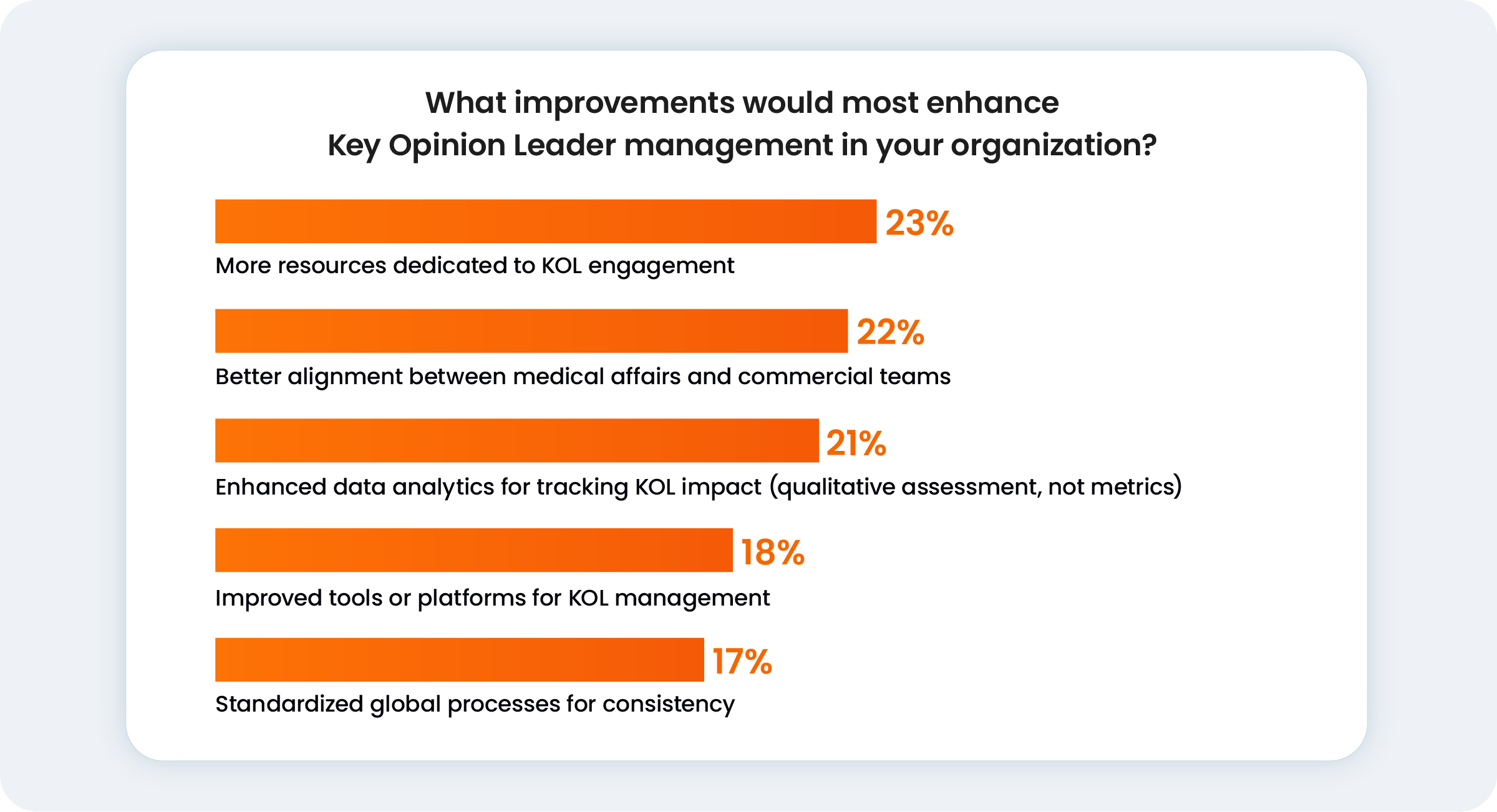
Beyond the technology, this fragmentation blocks the cross-departmental insights required for R&D and commercial success. For most medical leaders, achieving the necessary speed and compliance is now a balancing act between resource allocation (23%), internal alignment (22%), and upgraded data capabilities.
The Operational Cost of Data Fragmentation
While the path to modernization is a complex balancing act, the cost of the status quo is high. Operating with bad or disconnected data does more than slow down internal workflows—it creates a ripple effect of missed milestones. From delayed concept validation to stalled patient recruitment for clinical trials, these inefficiencies ultimately postpone the delivery of life-improving technologies to the patients who need them most.

The risks of poor data integrity can be categorized into two primary areas:

For medtech companies, medical congresses represent a high-velocity window for engagement. However, without a unified data infrastructure, teams often miss the mark. Success in this environment requires the ability to:
The move toward a unified ecosystem is about more than technical convenience; it is about organizational agility. Whether managing a global congress or preparing for an AI-driven future, the gap between leaders with “connected” data and those with fragmented systems is widening.
Unlocking the Potential of “Deep Data”
To overcome the risks of fragmentation, medtech organizations must shift toward “deep data”—information that is rigorously validated, structured, and globally connected. When teams have a single source of truth they can trust, they spend less time questioning the data and more time building scientific relationships.

As medical affairs evolves into a primary business driver, its influence extends across the entire product value chain – from initial design to market adoption. Unifying KOL management is the only way to streamline these cross-functional processes and ensure the rest of the organization can innovate based on deep, scientific insights.
Benefits of a Unified Data Foundation
A unified data architecture eliminates manual processing delays, enabling teams to transition from basic information gathering to high-impact strategic execution.
Ultimately, the transition to validated, well-structured data allows medical affairs to shift focus from the labor of gathering information to the power of making smarter decisions. For example, when exploring new research, combining KOL data with AI allows a team to instantly categorize medical topics based on impact—moving from data collection to strategic action in a fraction of the time.
Future-Proofing the Strategy
In an increasingly competitive market, medtech organizations can no longer afford to let poor data quality stand in the way of progress. Incomplete or siloed data is a liability that makes it difficult to act with certainty, leading to wasted resources and delayed market entry for new technologies. Turning this liability into an asset requires a shift toward data connectivity.
As AI continues to change the industry, data connectivity will be the primary factor in an organization’s success. Establishing a unified data foundation is a practical step toward ensuring that advanced analytics and future innovations are built on a reliable base. At its core, prioritizing data integrity is a commitment to the accuracy and speed required to deliver better outcomes for patients.
See how deep data from Link Key People can surface relevant insights and improve customer engagement.