Emerging Medtechs Focus on Improving Data Collection and Site Collaboration with Strategic Digital Support
Benchmark report illustrates unique strategic approaches for emerging medtechs
By Erich McGilvreay, Director of Clinical Strategy, Veeva MedTech
Macroeconomic pressures, shifting investment strategies, and more stringent data requirements like those under the EU MDR are changing the medtech clinical trial landscape. For emerging medtechs, these challenges are magnified by increased pressure to bring products to market quickly and efficiently, while operating with limited budget and resources.
The 2025 Veeva MedTech Clinical Benchmark Report provides insights from medical device and IVD companies on how they are prioritizing data collection and reconciliation, strengthening site relationships, and introducing processes to streamline operations for scale.
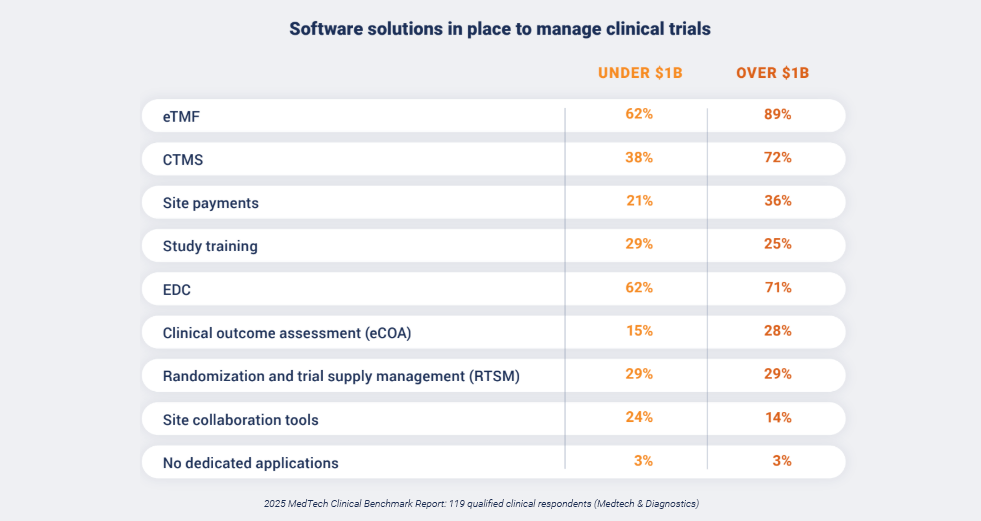
Data from the report highlights distinct differences where emerging companies (under $1B in revenue) are investing compared to enterprise and global companies (over $1B in revenue).
Navigating complex data requirements with minimal resources
More complex data requirements and a wider range of data sources are causing emerging medtechs to prioritize the implementation of an EDC system as the foundation for additional solutions like eTMF and CTMS. While a majority of survey respondents in the emerging category reported that they have adopted an eTMF, they lag behind enterprise and global companies (62% vs. 89%).
This is also the case for CTMS adoption (38% vs. 72%), but the two groups are nearly in-line with EDC adoption (62% vs. 71%). This initial focus on EDC is a pragmatic first step, as data collection and management is a common problem cited in medical device and IVD clinical trials.
The pressure to generate more comprehensive and continuous clinical evidence is intensifying. Site-entered EDC data remains the standard (82%) for the industry as a whole, but this evolving demand for other data sources increases the importance of investing in a robust data strategy and unified platform approach. Emerging medtechs prioritize site-entered EDC data and real-world evidence equally at 70%.
With 63% of emerging medtechs planning to optimize data collection and cleaning in the next year, they are making upfront investments in digital solutions and process improvements.
By harmonizing clinical systems and processes, companies can maintain a more holistic view of various data sources, site engagement, and the overall patient journey throughout the trial, while alleviating the administrative burden and resource limitations for all stakeholders.
Enabling sites to improve data collection and cleaning
In addition to a comprehensive data management strategy, companies are looking to optimize site recruitment and engagement. Site management is among the most resource-intensive processes, and delays can impact trial timelines, cost, and speed to market.
Site set-up, identification, and selection is cited as the biggest challenge during clinical trial startup (37%). This is often because emerging medtechs have less established site networks and limited resources to create a standardized and repeatable process. This challenge is further compounded by competition for sites, unique regulatory requirements, extensive training requirements, and service providers not dedicating sufficient service for site startup.
The identification of study sites, exchange of documents and data, and monitoring and compliance (46%) is the second biggest challenge for emerging medtechs when collaborating with sites, only trailing on-time data entry and quality (51%).
This highlights a major gap: only 24% of smaller companies have implemented dedicated site collaboration tools, even though 68% of all medtech teams prioritize reducing manual processes in site collaboration.
Simplifying the onboarding experience for sites can make collaboration with sponsors more attractive and seamless.
By providing a unified clinical platform with streamlined workflows, emerging medtechs can reduce the administrative load on sites. In an industry where top-performing research sites can choose their studies, reducing friction is crucial to becoming a sponsor of choice and attracting the best partners.
As part of a platform strategy, leveraging a CTMS can alleviate site selection challenges by providing a central repository for reference and historical data on trial and site performance, allowing sponsors to identify the right sites faster and accelerate startup.
Investing in a site collaboration application that is connected to the CTMS can streamline data and document exchange to help automate repetitive tasks, reduce manual effort, and eliminate the risk of data errors.
Closing the digital gap for long-term growth
Long-term success for emerging medtechs will require a balance between maintaining lean operations, generating necessary clinical evidence, and investing in scalable systems and process improvements.
Pragmatic investments in the right solutions at the right time with a cohesive strategy to maximize adoption and efficacy allows companies to solve key challenges today while scaling for tomorrow. While adopting end-to-end technology solutions is imperative, streamlining processes and investing in the right personnel will help yield higher returns on your technology investment.
A unified foundation for long-term success will help navigate changing regulatory requirements, macroeconomic trends, and clinical evidence demands, ultimately accelerating product approval. To navigate this landscape, companies can leverage a unified clinical technology foundation to help:
- Harmonize data with compliance and innovation in mind
- Build a connected digital ecosystem for 360-degree clinical trial management
- Focus on real-time and historical metrics to create a business case for future innovation
- Reduce the administrative burden on sites to develop stronger partnerships
Connecting data, documents, and processes across the clinical lifecycle provides the visibility and efficiency needed to make proactive decisions, accelerate study timelines, and ensure a continuous flow of high-quality data. Creating a unified technology foundation will also help prepare for AI implementation, an area which 72% of survey respondents plan to invest in over the next 12 months.
By establishing a unified foundation, emerging medtechs can satisfy rigorous regulatory standards and create a robust, future-proof structure needed to lead innovation for patients.
For additional insights, read the full report.