Rethinking How Claims Can Drive a Competitive Advantage in Medtech
While often viewed through a compliance lens, medtech claims represent the interface between innovation and commercial viability. A robust claims portfolio synthesizes clinical evidence, safety data, and market access requirements into a coherent value proposition and transforms regulatory adherence into competitive willingness-to-pay.
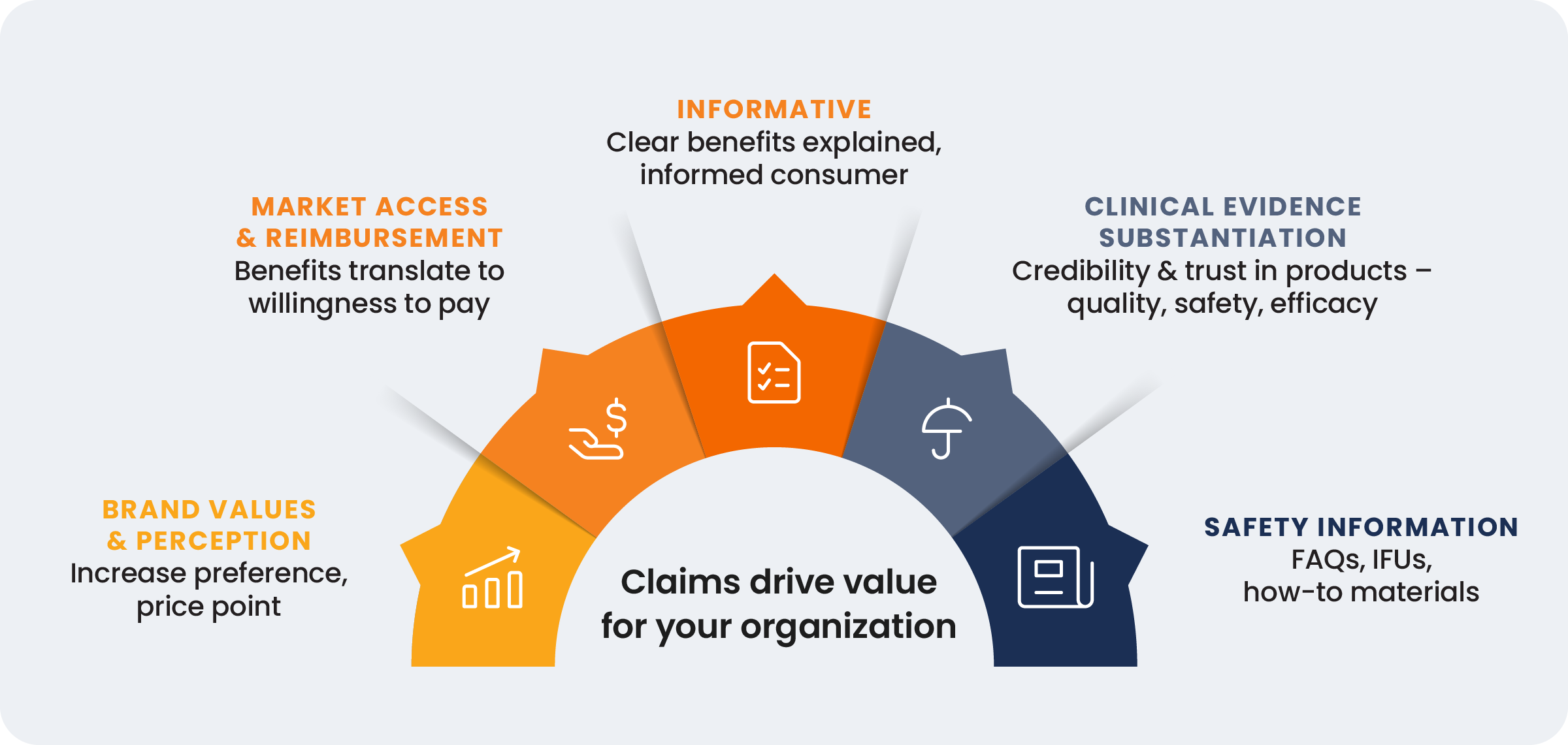
By integrating the traditionally siloed functions of regulatory, medical, and commercial, medtech organizations can better position themselves to win. A well-designed operating model provides the vehicle for success, but a unified, collaborative culture is the fuel that powers it. Without this cultural foundation, even the most sophisticated processes fail to deliver their full potential.
At a recent Veeva Medtech Summit, leaders from three world-class organizations, Coloplast, EssilorLuxottica, and Fresenius Medical Care, shared their collective claims experience, revealing that a unified mindset is the definitive characteristic that separates market-leading medtech organizations from the rest.
Silos dilute market impact
To unlock the strategic value of claims, organizations must first understand the high costs associated with a fragmented process. These costs are linked to operational inefficiencies with a tangible loss in speed to market, message clarity, and competitive sharpness. The most common point of failure is a deep, structural disconnect between the teams responsible for creating claims and the teams responsible for deploying them.
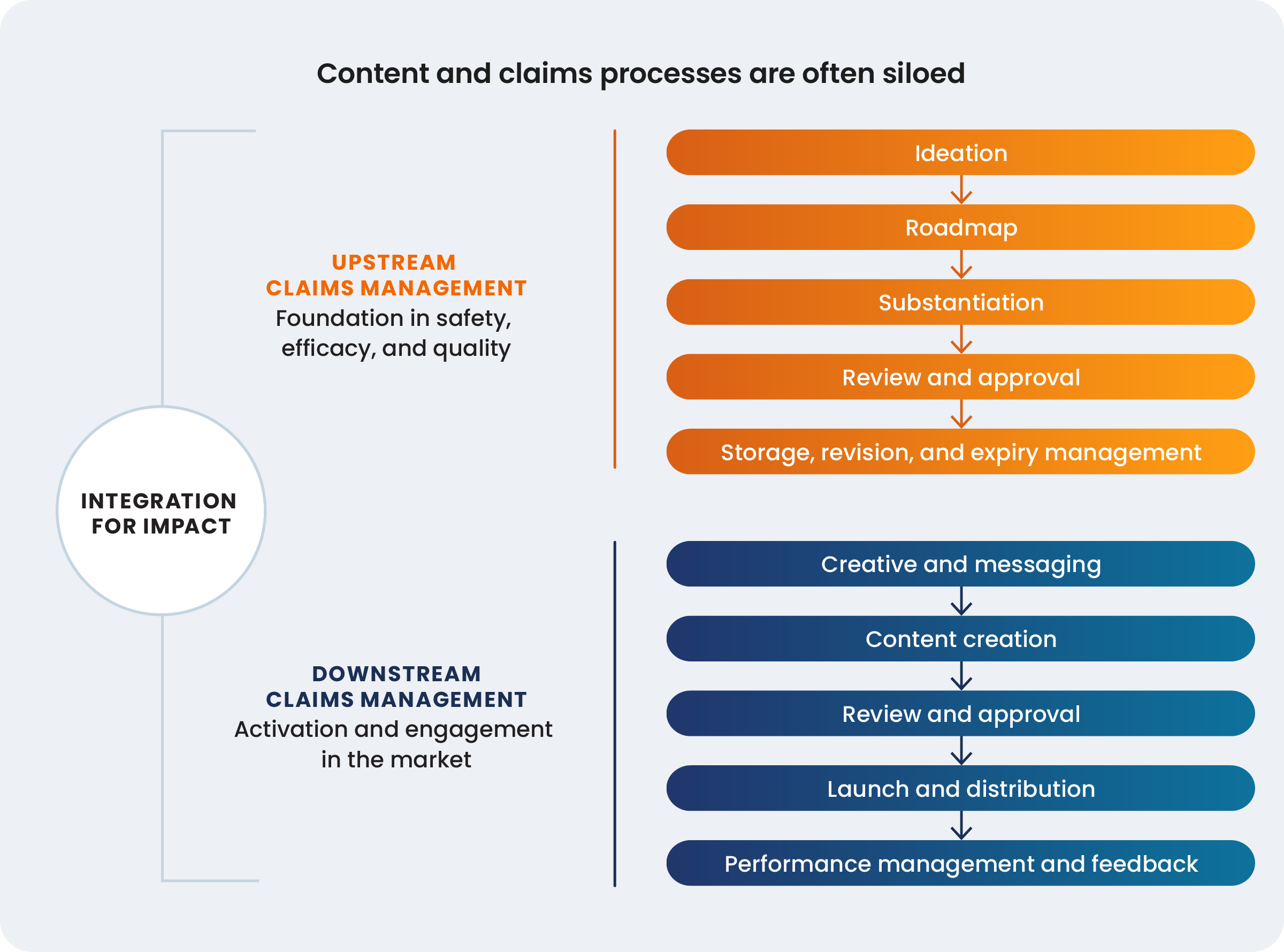
Findings from the 2024 Veeva MedTech Commercial Benchmark Report
In most medtech organizations, there is a clear upstream R&D process in which claims are ideated, substantiated with evidence, reviewed, and approved, often existing in a different universe from the downstream execution by marketing teams. This separation creates significant cultural friction. Ben Rochette, VP of global regulatory affairs at Coloplast powerfully articulated this dynamic with a simple metaphor: regulatory and medical teams can feel stuck in exaggerated stereotypes of being like the “grumpy uncle” at the family dinner when working with downstream marketing teams.
This perception is more than an internal dynamic and highlights a key business problem that stifles innovation and slows momentum. When regulatory is seen as the blocker and commercial is viewed as reckless, a “team A versus team B” mentality takes hold. This adversarial posture is a hidden tax on innovation, paid for with delayed launches, watered-down messaging, and market hesitation. While the team is debating internally, a more unified competitor is launching. Overcoming this cultural divide is the first step toward building market advantage.
Industry leaders identify two key things that power a winning claims engine
- 1. Designing claims to win from day one
By defining early stage claims, companies ensure that R&D investments are linked to high-value market outcomes. This approach also preempts usual late-stage technical debt and secures the scientific authority required to drive rapid adoption and command market share at launch.
Dimple Zala, Global Medical Advisor at EssilorLuxottica, emphasized that evidence must be treated as a purpose-built asset designed to secure a specific competitive advantage. “Instead of gathering evidence for the sake of gathering it or for its own sake, really what we need to think about is proving what will essentially help us win,” says Zala.
- 2. Fostering “healthy tension” through early creative symbiosis
True commercial agility requires reimagining technical stakeholders as proactive creative partners. As Derrick Greene, director of marketing content and operations at Fresenius Medical Care noted, this necessitates “early, intensive, cross-functional collaboration” integrating regulatory, medical, and commercial perspectives at the ideation phase rather than the final review.
The objective is to leverage what Ben Rochette (Coloplast) describes as “healthy tension.” By pressure-testing claims early, teams ensure they are both defensible and impactful, recognizing that sometimes friction between functions drives better market outcomes. Ben explained, “The collaboration comes with a sense of healthy tension across departments. We don’t have to agree on everything as we seek consensus, as long as we move together toward the same goal.”
Clearly a symbiosis emerges in which commercial insights shape the evidence strategy, while medical and regulatory expertise informs messaging to ensure it is credible and durable. Moving away from the external reviewer model allows technical partners to offer alternative pathways and creative solutions, improving regulatory guidance into a calibrated, predictable driver of productivity rather than a late-stage bottleneck.
Scorecard for claims impact
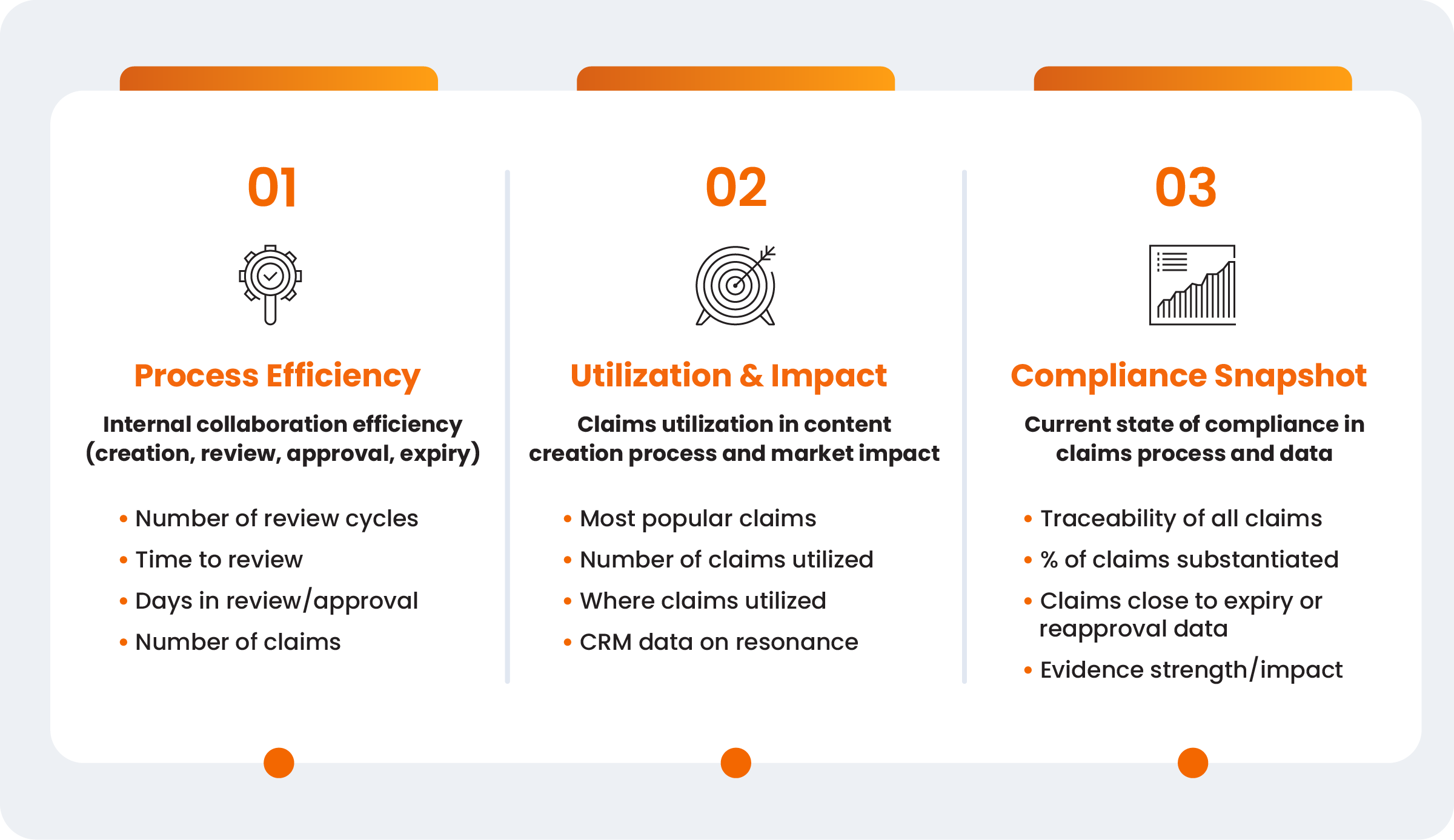
Most organizations don’t have insight into how their claims are helping them in the market, which claims are having impact, or how their internal operations could provide better support. Leaders agreed that while directly linking a single claim to a specific revenue figure remains “the dream,” a mature claims organization must move beyond vanity metrics to focus on three simple areas to start:
- 1. Process efficiency: Evaluates operational speed. A high number of review cycles serves as data-driven evidence of a lack of early alignment and higher risk in the market.
- 2. Utilization: Tracks a claim’s relevance and adoption. Low utilization in promotional material signals that a claim, regardless of substantiation, is not commercially relevant or is under utilized by the team.
- 3.Compliance snapshot: Monitors the library’s health, including traceability to evidence and upcoming expiration dates to manage organizational risk.
As Ben Rochette (Coloplast) noted, KPIs are essential for elevating conversations beyond subjective complaints (“it’s taking too long”) to a data-driven discussion about true organizational pain points. This objective data allows leaders to pinpoint where the process is breaking down and make targeted improvements, paving the way for the advanced capabilities that will define the future of claims management.
The Future of Claims Management
Organizations that succeed over the next five years will invest strategically in cultural and operational transformation. Panel leaders identified three priorities:
Mastering agility
Success requires cross-functional alignment on phased implementation. Winning organizations “start small, but aim big” with clear milestones and shared strategic vision. Misalignment, where commercial teams sprint while regulatory prepares, derails progress.
Achieving end-to-end consistency
Standardizing processes, roles, and responsibilities across the claims operating model creates a predictable engine that delivers consistent speed to market and eliminates operational volatility.
Building a “living claims ecosystem”
The most transformative vision involves evolving from static claims libraries to dynamic, continuously learning systems. By integrating real-world data and stakeholder insights, organizations can detect market shifts in real-time. AI serves as a key enabler and with near-term applications in competitive intelligence and compliance verification, it makes this vision actionable today.
To learn more about how Coloplast encourages its regulatory teams to view claims management through a commercial lens watch this video.