Veeva Vault CTMS
Manage Trials End-to-End
with a Modern CTMS
Proactively manage trials and accelerate toward
a more connected digital future.
选择VAULT CTMS的原因
提高效率
提高生产力
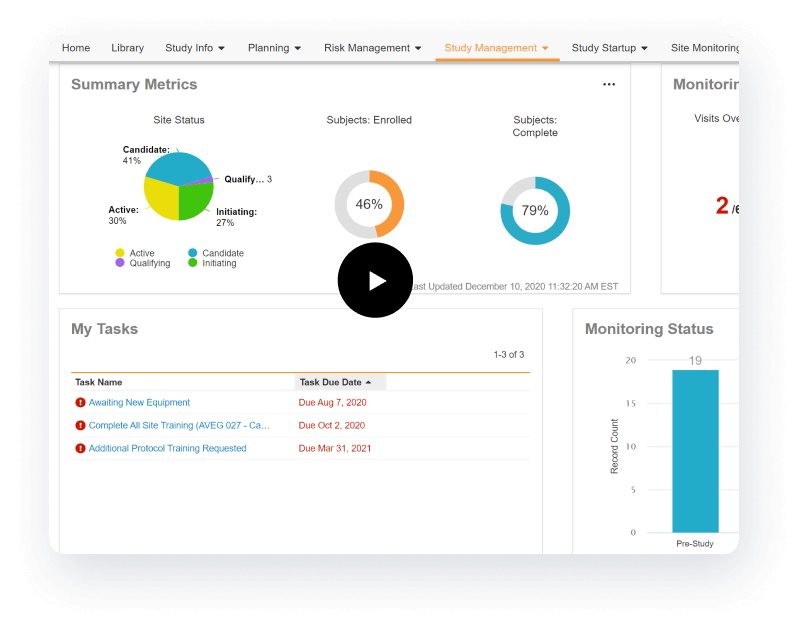
为研究团队配备基于角色的仪表盘和直观导航。改善决策
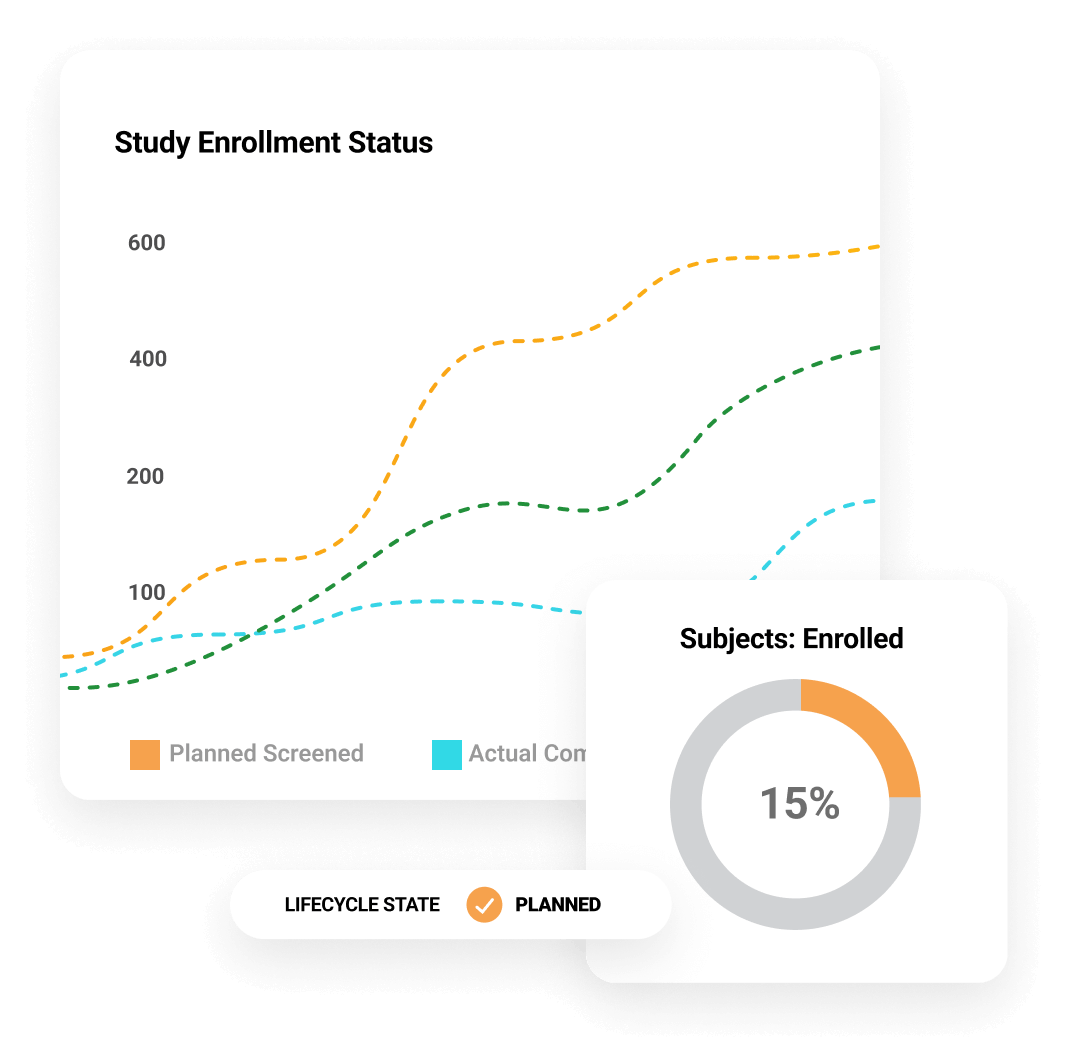
通过实时查看试验状态,实现闭环问题管理和战略规划。加快试验执行速度
主动识别和管理风险,减少时间延误。
提高效率
30%
提高监查规划和执行的工作效率
25%
提升问题管理效率
465
每1,000份访视报告可节省的天数