Optimize the validation lifecycle
Announced 2021
Status Mature
Customers 11-50
Veeva Validation Management streamlines the qualification and validation process for computer systems, facilities, utilities, equipment, and processes.
Track system inventory, requirements, and project deliverables in one digital solution to improve oversight and simplify validation efforts.
Easily create validation activities, execute test scripts digitally, and generate summary reports all within a single system designed to enhance efficiency and support compliance.
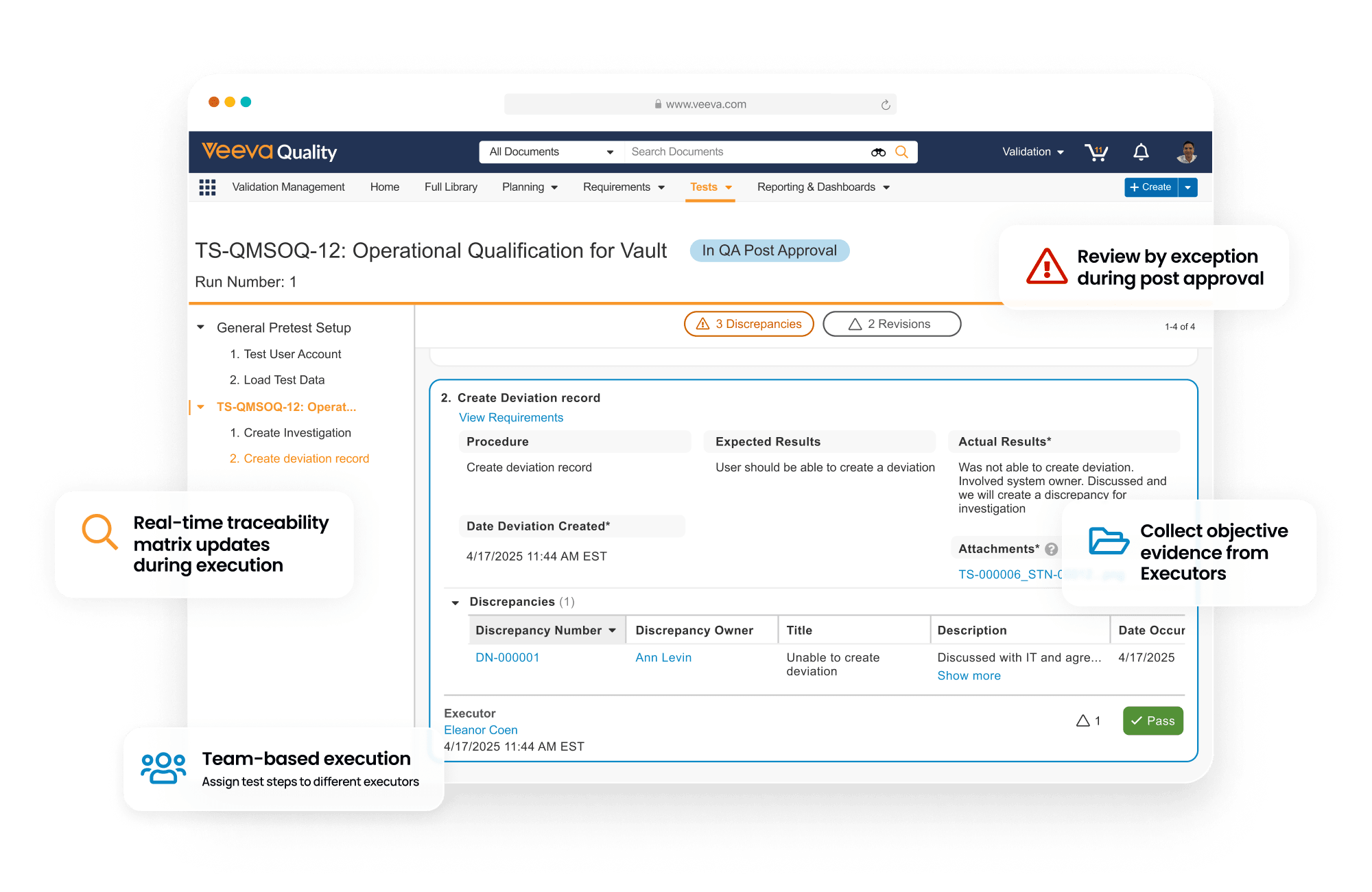
Veeva Validation Management Impact
Improve validation efficiency
Reduce bottlenecks with automated workflows for commissioning, qualification, and validation across systems, facilities, utilities, equipment, and processes.Deliver faster, more accurate results
Accelerate validation with simplified inventory tracking, standardized requirements, and streamlined project deliverables. Digitally execute test scripts and auto-generate traceability matrices and summary reports.Unify validation and quality
Ensure compliance with connected workflows, real-time reporting, and continuous validation across systems, equipment, processes, and sites on a unified platform.Enhance traceability and data accuracy
Automate traceability and extend capabilities with Veeva API to support business-specific requirements. Ensure higher quality data is accessible for risk management, analysis, automation, and AI.
See Veeva Validation Management in action
Customer Success
Medtechs unify quality management with Veeva Quality










Read article
Thermo Fisher Scientific shares insights from its multi-year effort to instill a culture of quality

View infographic
Insights from industry leaders on how the industry is thinking about proactive quality management
Read brief
Veeva Validation Management Features Brief

Watch video
NorrDia streamlines audit readiness and tracks key quality metrics with Veeva Quality Cloud



