COMPLIANCE REPORTING
Visibility into read and understood, periodic review, and other workflows allowing easy reporting of outstanding or upcoming tasks, and mitigating compliance risks.
CONTROLLED CONTENT ACCESS AND COPY DISTRIBUTION
Securely access and use documents from any major web browser, or if enabled, print, distribute, and track controlled PDF copies. The printed document contains a configurable overlay displaying information such as the person who downloaded it, when it was downloaded, and how long the copy is valid.
COMPREHENSIVE AUDIT TRAIL
Easily demonstrate compliance with detailed audit trails capturing every event in a document’s history, including document approvers and reviewers, status changes, execution of a signature, and more.
ELECTRONIC SIGNATURE AND MANIFESTATION
Approve documents using electronic signatures and manifestations that are compliant with Title 21 CFR Part 11 and Annex 11.
Learn More
Customer Story
DOCUMENT CHANGE CONTROL
Track and control document changes—such as withdrawal, approval of revised content, and release for use, automatically gathering proposed changes and reason for changes to support future audits.
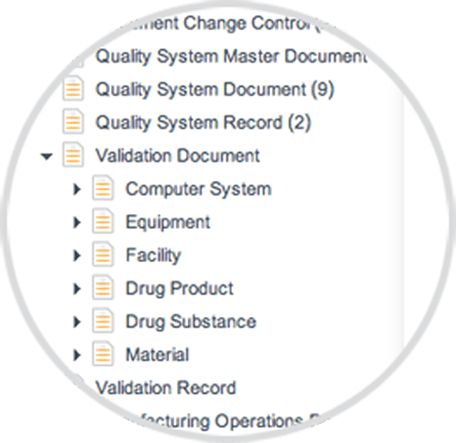
QUALITY DOCUMENT TAXONOMY
Predefined document taxonomy, metadata, and pick lists for quality, manufacturing, and validation documents facilitates operational harmonization, and allows organizations to quickly adopt best practices.
CONFIGURABLE QUALITY WORKFLOWS
Build in compliance with automated quality workflows including document expiration, periodic review notification, and training flags. Easily extend workflows to include quality management systems with our Sparta TrackWise Connector and to learning management systems to enable end-to-end quality processes.
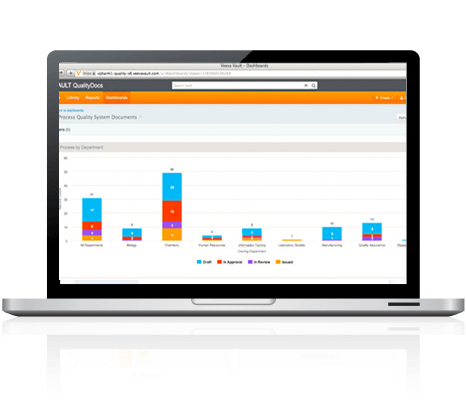
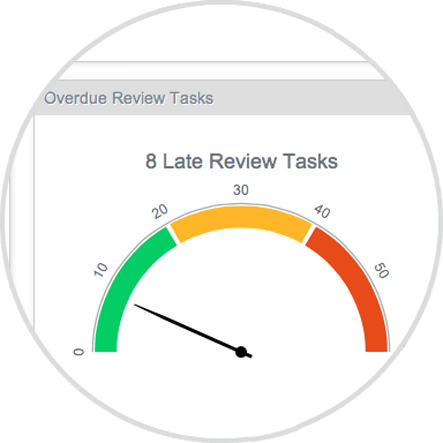
INTERACTIVE DASHBOARDS AND REPORTS
Self-service reporting and dashboards enable users to see status of content and processes, making it easy to identify bottlenecks, click through the report for more detail, and share the information with your team and partners.