Announced 2025
Status Available for Early Adopters
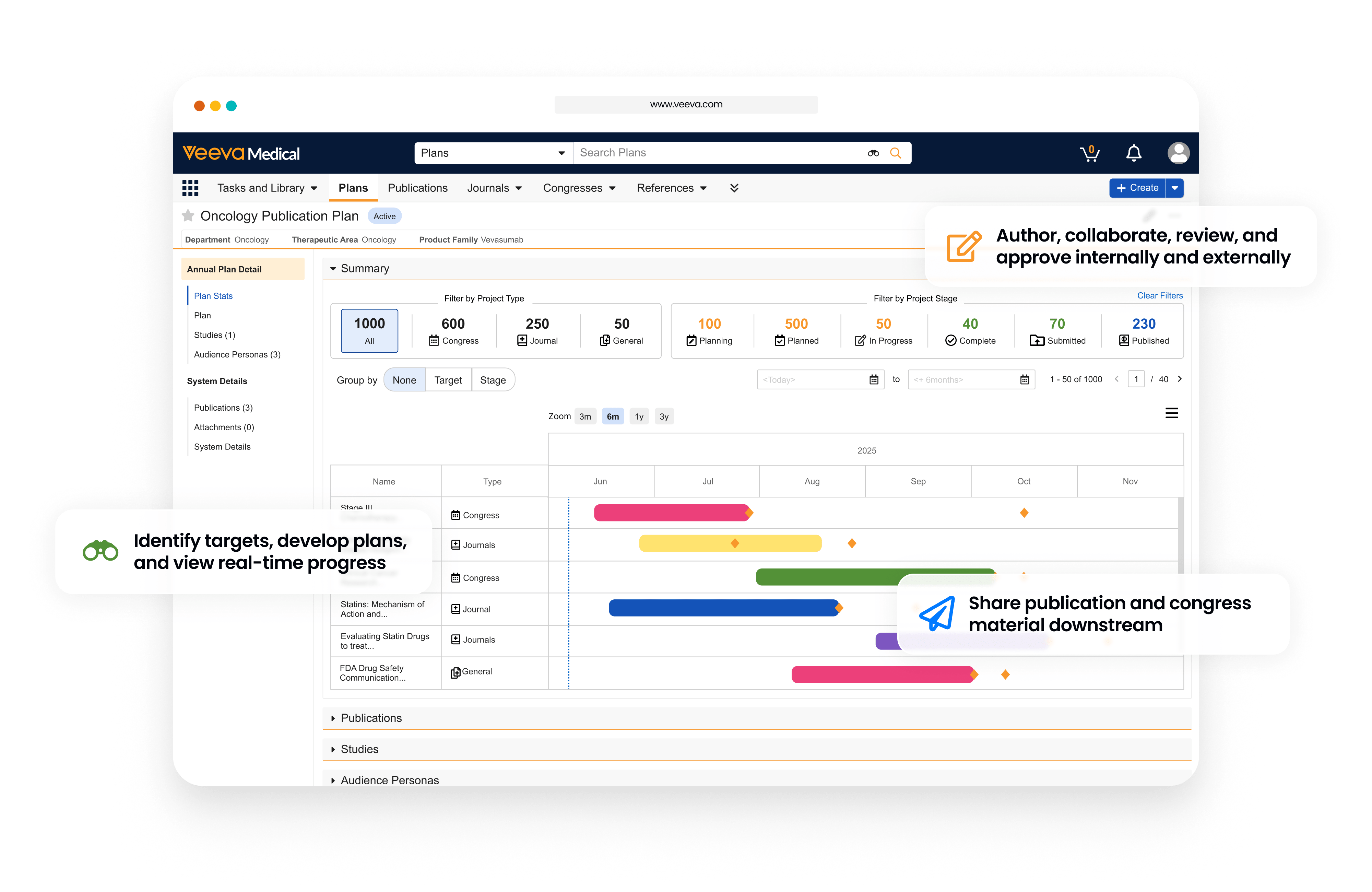
Simplify the End-to-End Publication Process
Veeva Publications is used to plan, author, review, approve, and submit scientific literature to journals and congresses.
Teams can develop publication plans and manage key milestones with tailored views by therapeutic area, publication type, and other categories.
Real-time collaborative authoring and task management allow internal teams and external authors to develop content seamlessly in a secure workspace.
Dashboards and reports provide complete visibility and the ability to quickly pull metrics on the publication pipeline and impact.
Publications connects with other Veeva medical applications to facilitate the flow of scientific evidence across systems.
Veeva Publications Impact
Strengthen Project Execution
Streamline planning across the portfolio and easily manage publications from strategy to submission.Simplify External Collaboration
Enable real-time collaboration with external contributors in a secure, intuitive shared workspace.Gain Full Visibility Across Publications
Utilize dashboards and reports to track publication reach and downstream impact.Be Compliant By Design
Ensure audit readiness with built-in compliance aligned to journal and regulatory requirements.
Customer Success
Medtechs unify scientific exchange with Veeva Medical






Rethink the Model for Publications Management in Medtech
Read White Paper
Industry Overview: The State of Medical Affairs in Medtech
Download Infographic
Expert Insights to Transform Your Evidence Generation Strategy
Read Article