Vault eTMF
Streamline document collection, management and analysis.
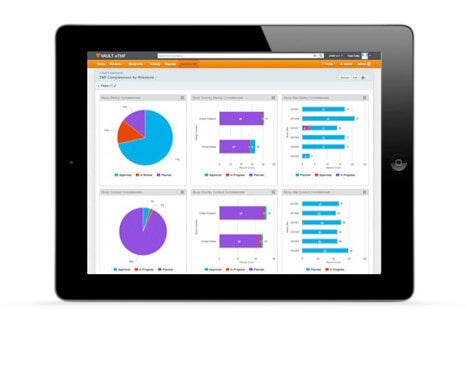
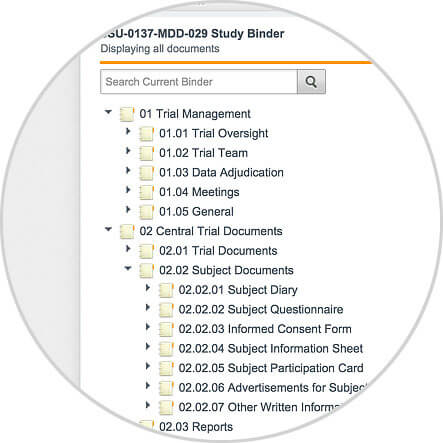
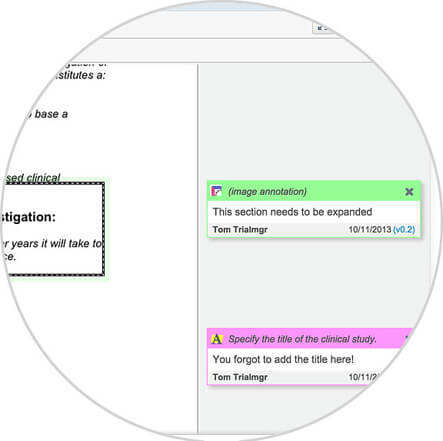
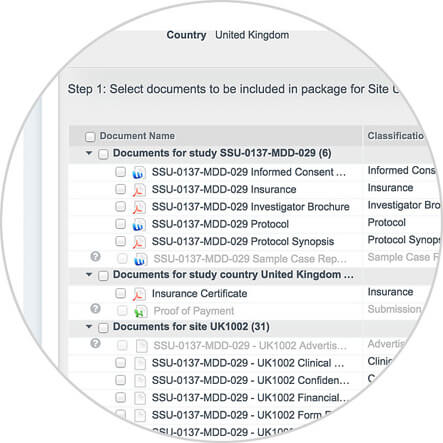
Take a quick look inside Vault eTMF to see how sponsors and CROs are able to easily manage their clinical trials, as well as create comprehensive business reports, by utilizing a modern, cloud-based electronic trial master file solution.
Contact Us