Resource Center
AI (Veeva AI)
View More AI (Veeva AI) ResourcesClinical Data
View More Clinical Data ResourcesLearn More
Survival of the Fastest: A Practical Framework for Risk-Based Data Management

Watch Demo
Veeva CDB Feature Highlights: Protocol Deviations and More
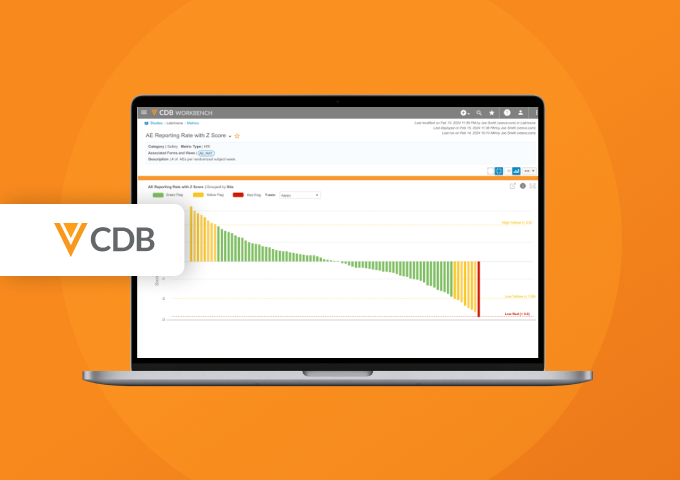
Watch Video
Boehringer Ingelheim: Unified Clinical Platform

Watch Video
Boehringer Ingelheim: Clinical Data One-Stop Shop

Clinical Operations
View More Clinical Operations ResourcesLearn More
Recordati Passes Recent Inspection with No Findings

Watch Video
Boehringer Ingelheim: Unified Clinical Operations Success

Learn More
Biopharma Speeds Trials with Unified Clinical Platform

Learn More
Lotus: Accelerating Trials with Automated eTMF

Commercial
View More Commercial ResourcesWatch Video
Keynote Highlights: Agentic Commercial™ and the Breakthrough of Commercial Evidence™

Watch Video
Commercial Evidence Generated from the Field with the Agentic Call Report™

Watch Video
Commercial Evidence from Conversational AI for Patients and HCPs with Ostro

Read Press Release
More than 125 Customers Worldwide Live on Vault CRM

Learn More
Novel Therapy Brands - Launch Performance Q1 2026

Watch Videos
Bayer: Powering Medical, Commercial, and Clinical Teams with Link Data

Learn More
Fragmented Data Is Holding Back Biopharma Launches

Learn More
BI: Global Data Harmonization with Veeva

Development Cloud
View More Development Cloud ResourcesRead Press Release
Veeva Announces Falcon, an Agentic Platform and Standard Agents to Deliver Agentic Labor in Drug Development

Read Press Release
AOP Health Standardizes on Veeva Industry Cloud for Life Sciences Across All Business Areas

Watch Demo
Quality-Safety Connection
Learn More
OM Pharma: Building a Data Foundation for Rapid Growth

Medical
View More Medical ResourcesQuality
View More Quality ResourcesLearn More
Automating Market Ship Decisions with Veeva Batch Release

Read Press Release
Veeva Quality Cloud Advances Operations for Kindeva

Learn More
The Essential Ingredient for GxP Training Innovation

Learn More
Accelerate Lead Times with External Collaboration in Quality

Regulatory
View More Regulatory ResourcesResearch Sites
View More Research Sites ResourcesLearn More
6 Strategies to Simplify Study Training for Sites

Read Press Release
Veeva Announces eSource Application to Eliminate Paper

Read Press Release
Veeva and SCRI Form Strategic Collaboration

Read Press Release
Veeva Announces Research Site CTMS

Vault Platform
View More Vault Platform ResourcesRead Press Release
Teva Pharmaceuticals Commits to Veeva Vault CRM

Learn More
Copying Vault Data Using Direct Data API and Migration Mode

Watch Demo
Safety-EDC Connection Automates SAE Intake
Watch Demo
Safety-Clinical Operations Connection Automates Safety Letter Distribution
Result Not Found
The query that you've entered is not found.















