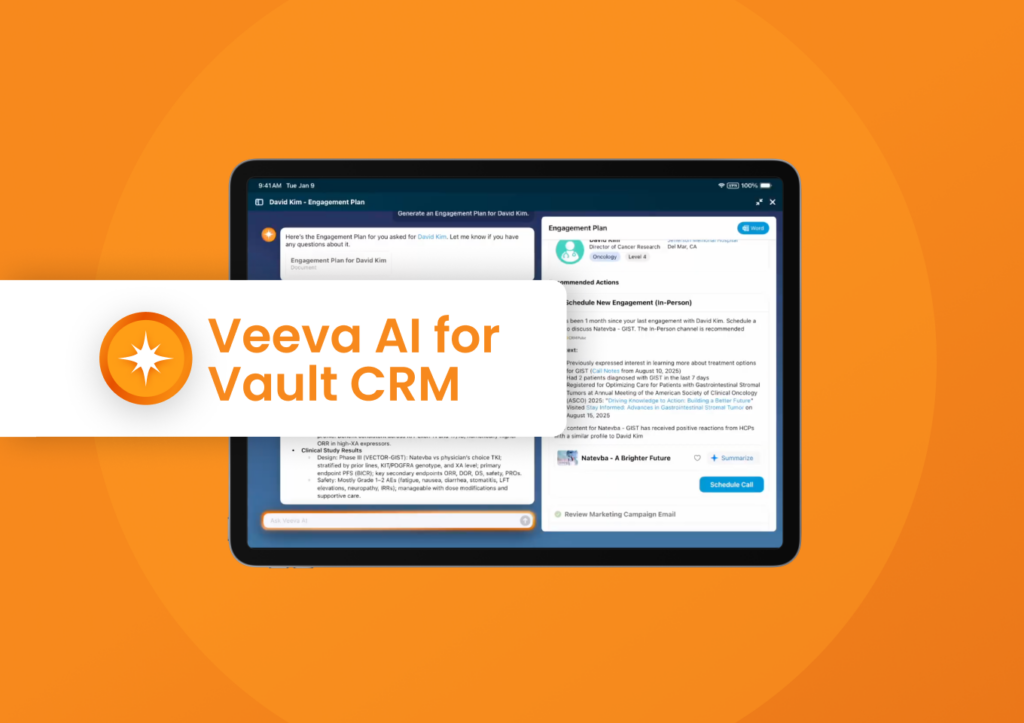
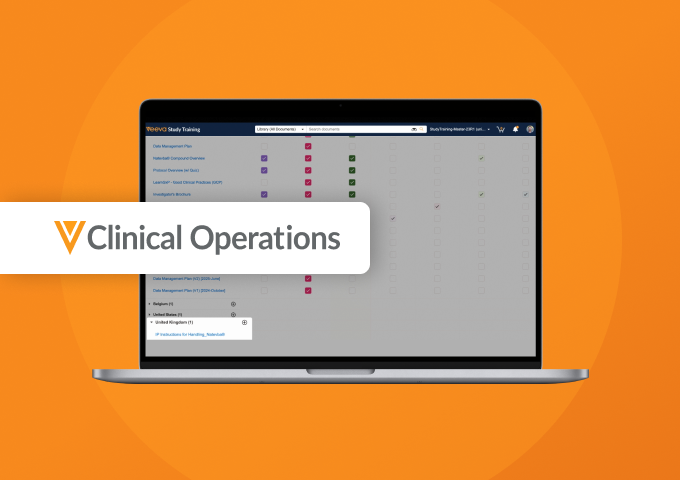
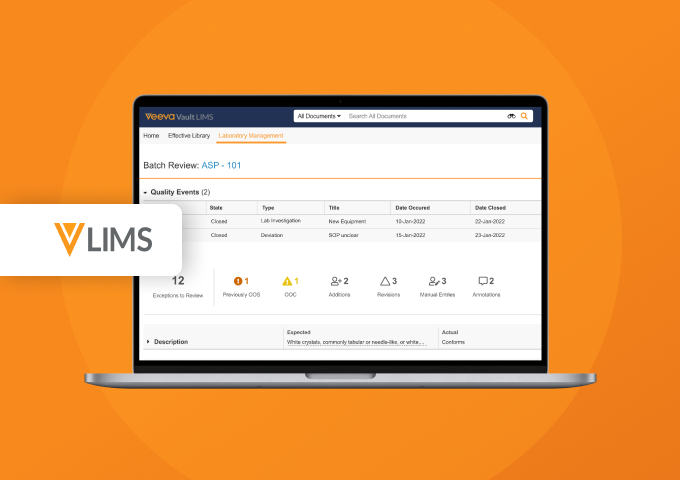
Demo Center
Watch video demos of Veeva applications, agents, and data solutions.
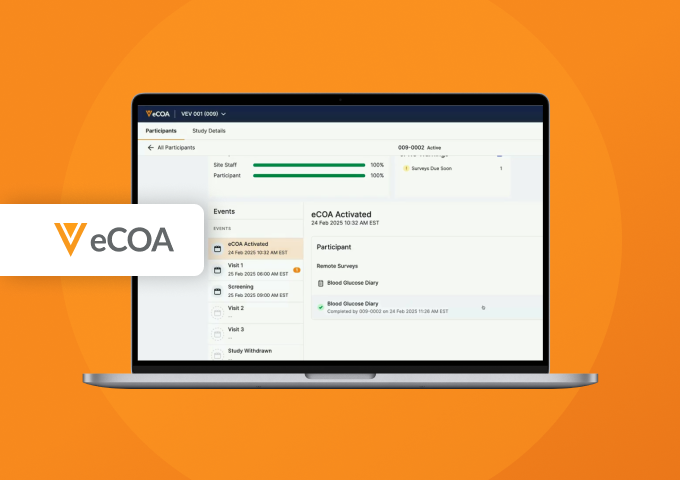
RTSM-eCOA Connection

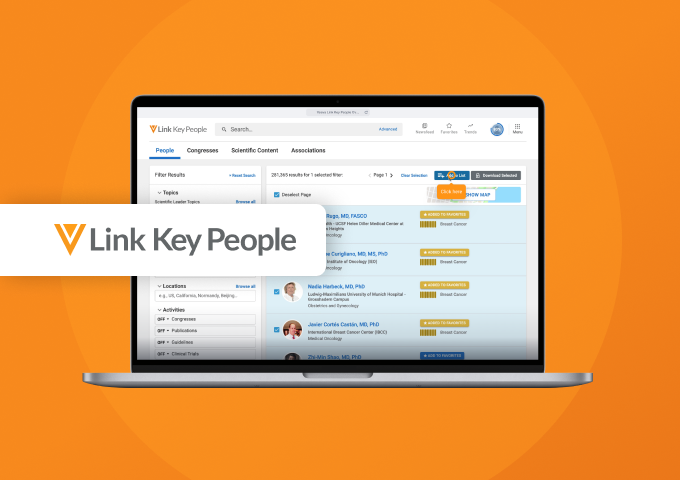
Link Key People in CRM

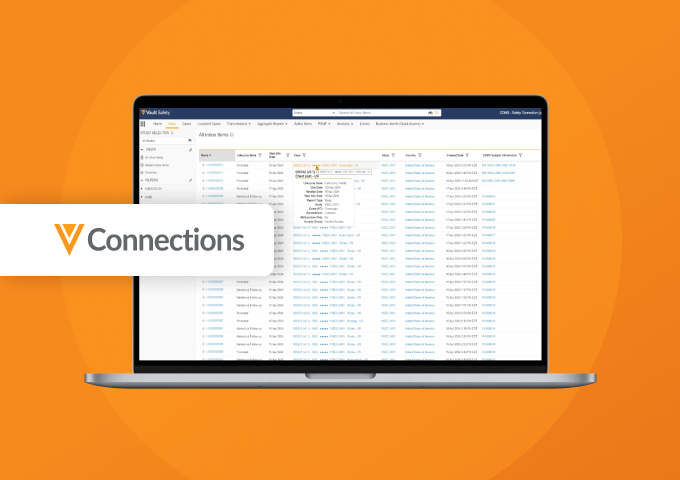
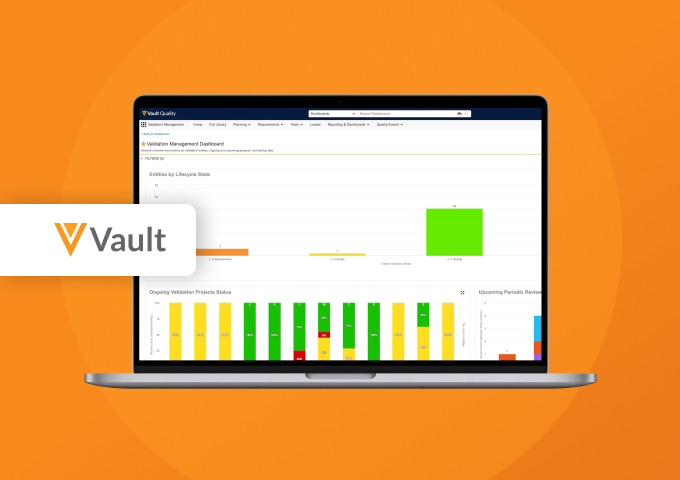
Quality-Safety Connection
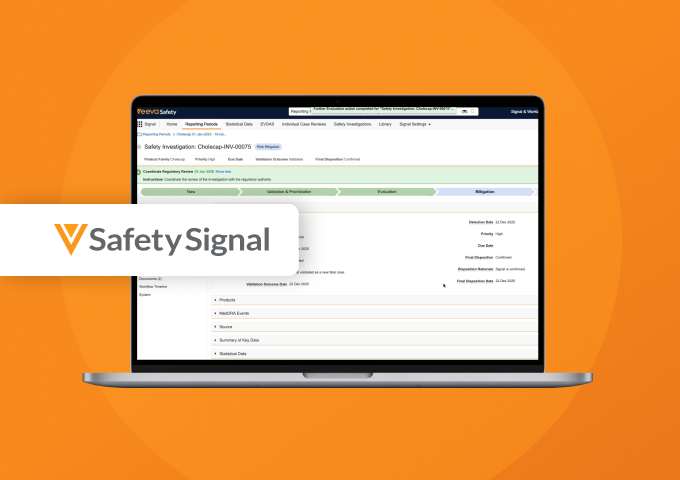
Veeva Safety Signal
Case Intake Agent and Case Narrative Agent

Global and Japan Case Processing in One Solution
Centrally Manage Safety Content and Processes
Simplify Signal Management and Safety Investigations
Application Assistant Agent

Health Authority Interactions Agents

Veeva RIM End-to-End Platform

Veeva Registrations

Veeva Submissions
Veeva Submissions Publishing

Veeva Submissions Archive

Veeva Publishing eLearning

Medical Solutions

More Relevant Engagement with Veeva Link Key People
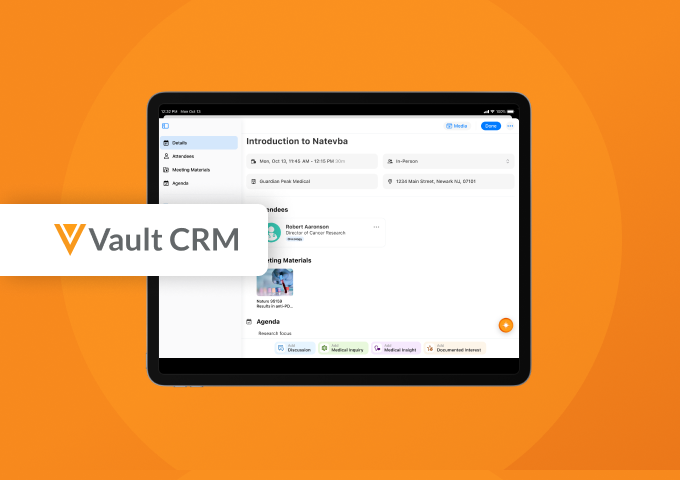
Reimagining Engagement with Medical Interaction in Vault CRM
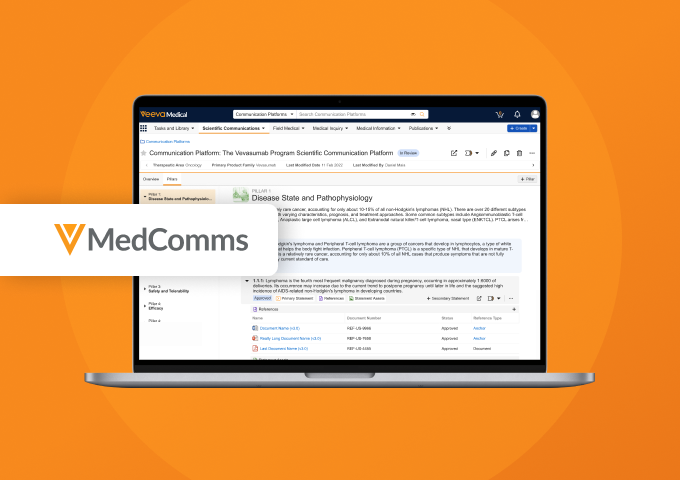
Scientific Communication Platforms in Veeva MedComms
Result Not Found
The query you've entered was not found.