Features Brief
Veeva Publications Features Brief
Veeva Publications unifies the end-to-end publication lifecycle from planning through submission to journals and congresses.
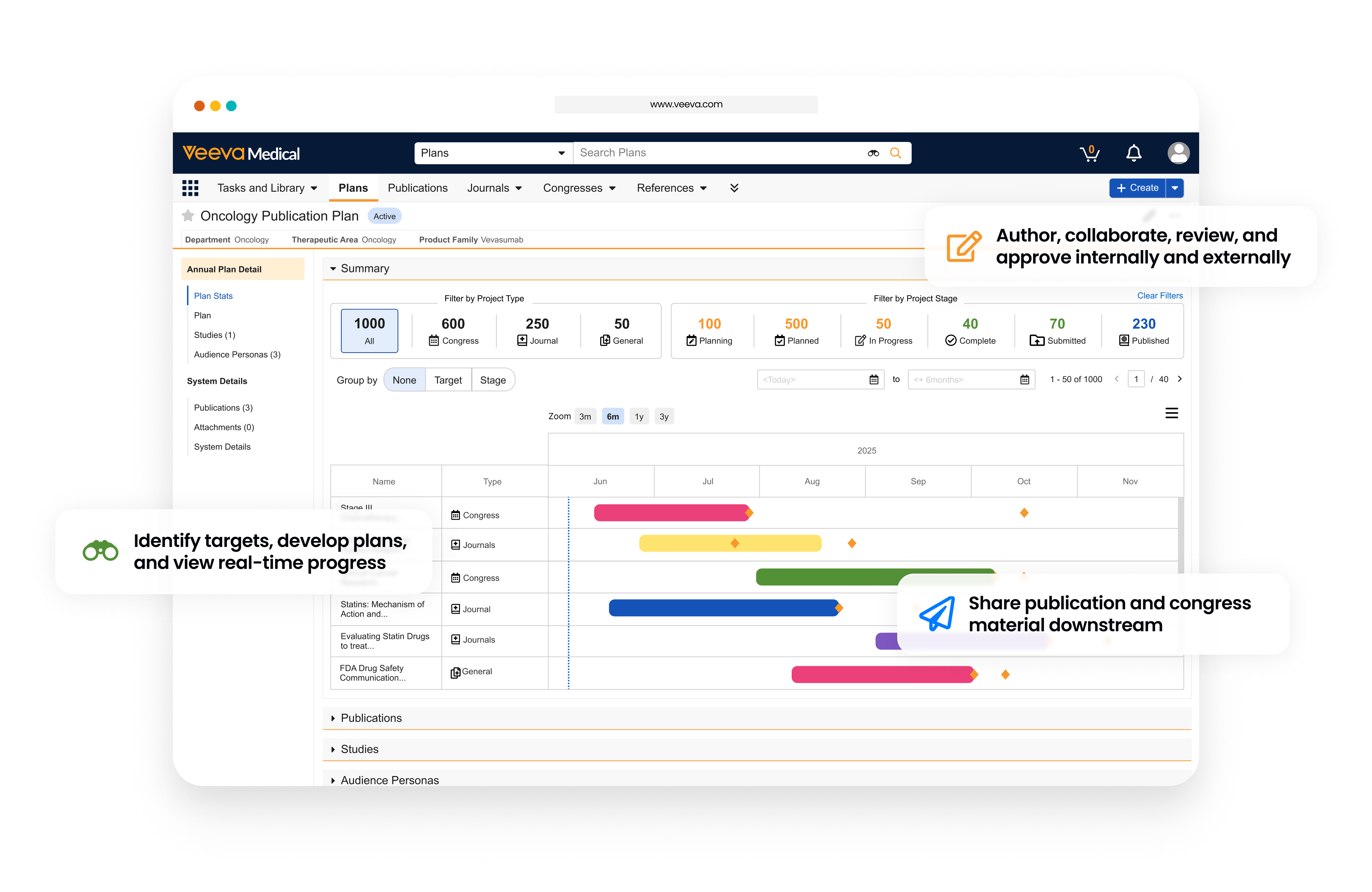
Unify the Publications Management Lifecycle

Publications transform complex data into the actionable findings that define how therapies are understood and trusted in a clinical setting. When planned strategically, publications accelerate the path from clinical study to scientific impact. However, common challenges across the lifecycle present a clear opportunity to improve efficiency and reduce manual steps.
Veeva Publications is a modern, intuitive solution that simplifies the end-to-end publication process for medical affairs teams. Built on the trusted Vault Platform, it enables organizations to plan, author, review, approve, and submit scientific literature to journals and congresses within a single, connected environment.
Business Benefits
-
Strengthen Project Execution
Streamline planning across the portfolio and easily manage publications from strategy to submission.
-
Simplify External Collaboration
Enable seamless access and real-time collaboration with external contributors in a secure, shared workspace.
-
Gain Full Visibility Across Publications
Utilize dashboards and reporting capabilities to track publication reach and scientific impact.
Key Features
-
Strategize and Plan
- Develop plans and project portfolios by therapeutic area, publication type, and more.
- Track key milestones with interactive Gantt Charts and configurable, personalized table views.
- Visualize projects and view real-time progress across adjustable timelines.
-
Create and Collaborate
- Bring internal and external contributors together in a single, connected, and secure workspace.
- Eliminate friction for external users with easy login access and an intuitive user experience.
- Enable real-time collaborative authoring and revision tracking with external contributors.
-
Review and Approve
- Track document versions, review notes, and author contributions automatically.
- Establish configurable workflows for coordinating review and approval tasks across stakeholders.
- Maintain transparency with robust version control and complete audit trails.
-
Submit and Track
- View detailed dashboards to get a clear picture of progress and performance across tasks.
- Pull pipeline metrics to see submission counts, target progress, and time-to-publication data.
- Measure downstream impact by tracking usage in approved scientific content in MedComms.
Connected ecosystem built on the Vault Platform
-
Leverages the latest cloud technology, is IQ/OQ validated, PQ ready and easily delivered through the web.
-
Part of Vault Medical, connects natively with MedComms, MedInquiry, and Grants (future) in a secure, high performance, validated environment.
-
With Veeva AI embedded in the Vault Platform, Agents operate with application-specific context, prompts, and safeguards, and direct, secure access to data, documents, and workflows.
-
Supports access to the wider medical ecosystem with an open, published API to easily integrate with other platforms and channels.
