Veeva CDB
Automate Clinical Data Workflows and Reconciliation
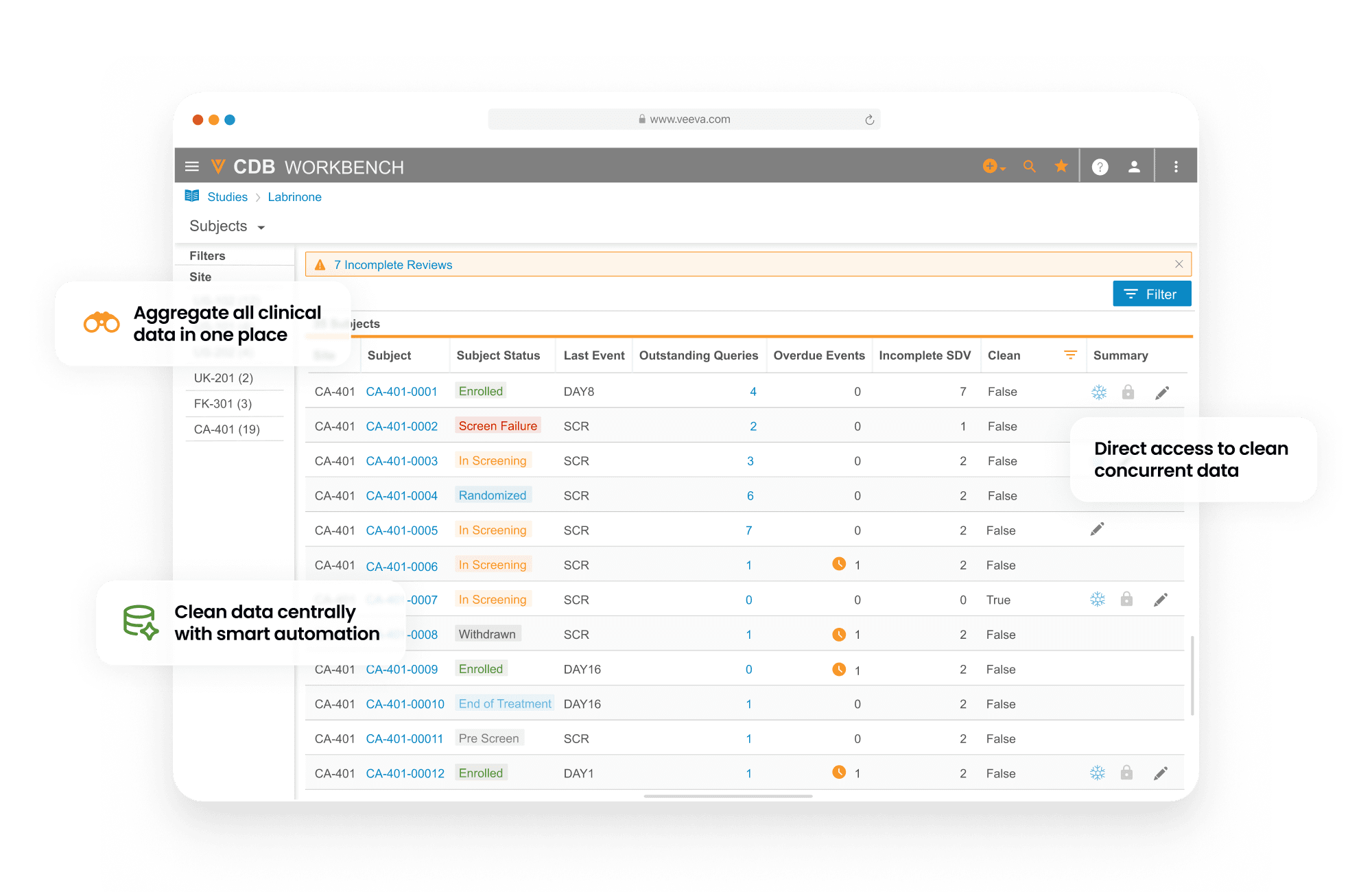
Veeva CDB provides a central environment to manage all trial data, automate workflows, and orchestrate review across the clinical ecosystem. The system automatically aggregates, reconciles, and cleans data from multiple sources—including EDCs, RTSM, eCOA, labs, imaging, and more—to provide a harmonized data package for downstream use.
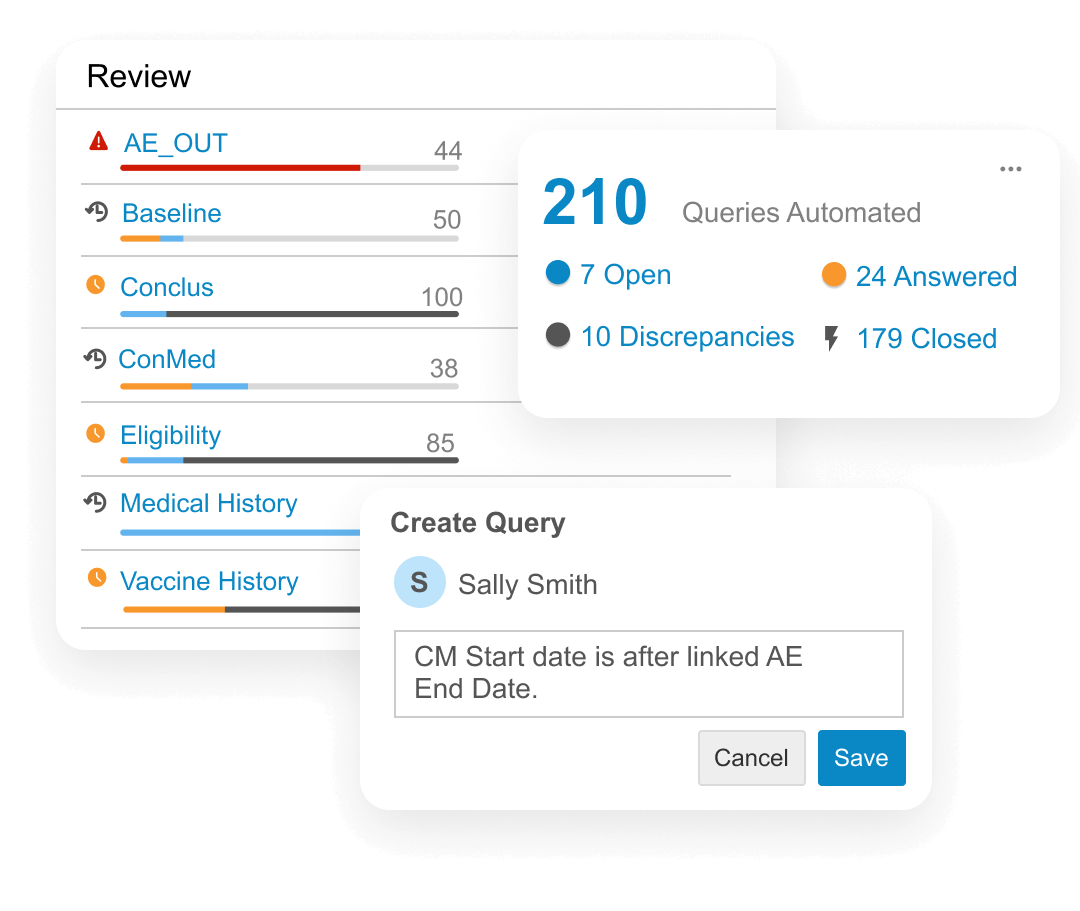
Data managers use CDB to create and manage queries, protocol deviations, and communications with data providers across all sources. The system automates data actions, including change detection and the creation or closure of queries and protocol deviations. Cross-functional teams collaborate on data review in real-time, while oversight teams monitor study health via interactive dashboards.
Announced 2018 Status Early Customers 11-50
Veeva Clinical Database Aggregates and Cleans Study Data up to 50% Faster
Overview
Automate Clinical Data Workflows and Reconciliation
Veeva CDB provides a central environment to manage all trial data, automate workflows, and orchestrate review across the clinical ecosystem. The system automatically aggregates, reconciles, and cleans data from multiple sources—including EDCs, RTSM, eCOA, labs, imaging, and more—to provide a harmonized data package for downstream use.
Data managers use CDB to create and manage queries, protocol deviations, and communications with data providers across all sources. The system automates data actions, including change detection and the creation or closure of queries and protocol deviations. Cross-functional teams collaborate on data review in real-time, while oversight teams monitor study health via interactive dashboards.
Impact
Exceed the likely outcome
30%
cleaning effort saved through automation
50%
easier to generate listings
78%
check queries automatically closed
Why Veeva CDB
Access complete and concurrent clinical data
Customer Success
Cuts time to aggregate
and clean study data by 30-50%











Resources
Explore and Learn
Watch Video
Boehringer Ingelheim Boosts Efficiency and Oversight with Veeva CDB

Read Features Brief
Veeva Clinical Database Features Brief

Read White Paper
Survival of the Fastest: A Practical Framework for Risk-Based Data Management

Read eBook
Clinical Data Workbenches: A Buyer’s Guide

Read Press Release
Veeva Clinical Database Crosses 200 Study Milestone

Watch Video
How a Top Biopharma Gets Quality Data Faster with Veeva CDB

Watch Demo
Veeva CDB Product Demo
Read Blog Post
Beyond the Assembly Line: Mastering the Elements of Clinical Data Science

Read Blog Post
Clearing the Technology Path to Clinical Data Science

Watch Video
4 Ways Smart Automation Accelerates Clinical Data Management

Watch Video
Syneos Health Transforms Clinical Data Management for Their Sponsor

