Resource Center
Clinical Data
View More Clinical Data ResourcesRead Press Release
Veeva Announces eSource Application to Eliminate Paper

Learn More
Clinical Data Managers and CRAs Warn of Data Quality Risks

Learn More
A Blueprint for Clinical Trial Transformation: Results from Three Top 20 Biopharmas

Watch Video
UCB Drives Patient Engagement and ROI with Veeva eCOA

Clinical Operations
View More Clinical Operations ResourcesLearn More
Recordati Passes Recent Inspection with No Findings

Learn More
Improve Site Relationships for Faster, Higher-Quality Trials

Learn More
Faster Training, Faster Trials: Guide for Site Training

Learn More
TMF Trend Report: 6 Trends of 2025

Commercial
View More Commercial ResourcesRead Press Release
More than 125 Customers Worldwide Live on Vault CRM

Watch Video
Italfarmaco: Accelerating Rare Disease Launch with Vault CRM

Learn More
Instant, Compliant Messaging with Vault CRM Engage

Learn More
Veeva AI for PromoMats

Medical
View More Medical ResourcesQuality
View More Quality ResourcesLearn More
AstraZeneca: Embedding Partners Into the Global Supply Chain

Learn More
Learn, Confirm, Then Scale

Learn More
Richter BioLogics Drives Efficiency and Compliance with Unified Quality

Watch Video
medac: Simplifying Training and Document Management

Regulatory
View More Regulatory ResourcesVault Platform
View More Vault Platform ResourcesWatch Video
Ethos: 4 Considerations for a Principled MLR in the AI Era

Watch Video
Sanofi: Unified Clinical Operations

Learn More
SMB Biopharma Perspectives on the Value and Impact of Vault CRM

Watch Video
Uncovering Critical Signals to Drive Strategic Action

Learn More
Boehringer Ingelheim Is a Veeva AI Early Adopter

Watch Video
Boehringer Ingelheim: Unified Clinical Platform

Watch Video
Boehringer Ingelheim: Clinical Data One-Stop Shop

Learn More
Free Text in CRM: A Legal and Compliance Guide

Learn More
The Role of Medical Affairs in Times of AI

Watch Video
Bayer AG’s Data Foundation for AI

Watch Demo
Pre-call Agent in Vault CRM
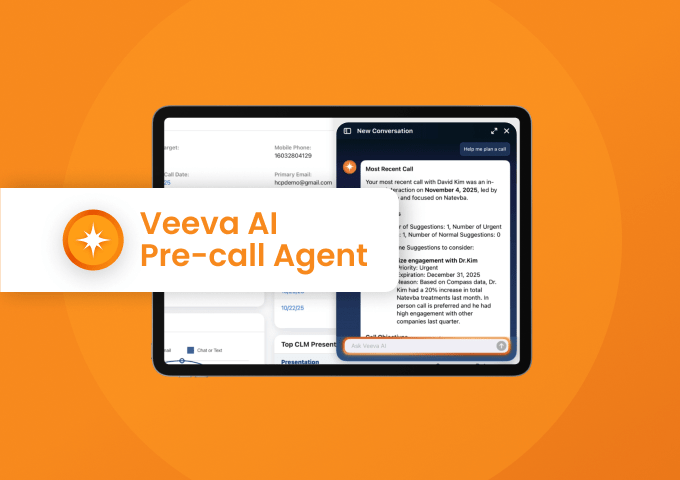
Watch Demo
Free Text Agent in Vault CRM

Watch Demo
Voice Agent in Vault CRM
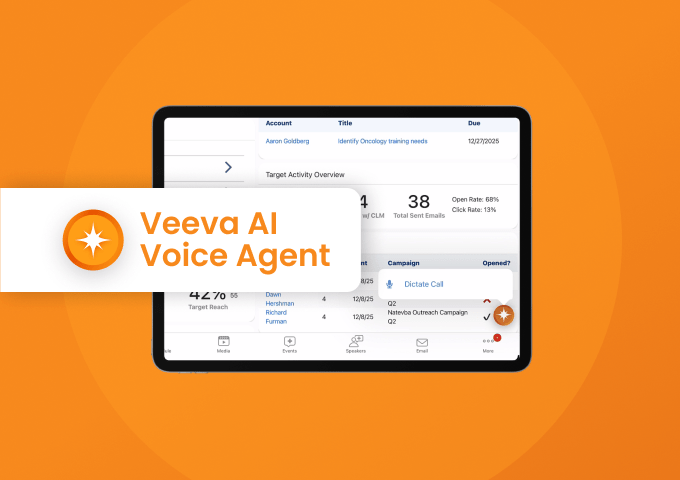
Watch Demo
Media Agent in Vault CRM

Learn More
RTSM-EDC Connection

Learn More
RTSM-eCOA Connection

Watch Video
Sandoz Increases Efficiency by Harmonizing Data

Watch Video
UCB Gains Efficiency with Unified Safety System

Watch Video
Boehringer Ingelheim Boosts Efficiency with Veeva CDB

Learn More
6 Strategies to Simplify Study Training for Sites

Result Not Found
The query that you've entered is not found.















![Safety [R]Evolution Podcast](https://www.veeva.com/eu/wp-content/uploads/2025/12/resource-tile-safety-revolution-340x240-1.png)



