Blog
Unlocking Clinical Data Value for Patients, Sites, and Sponsors
May 19, 2026 | angela.maffei@veeva.com
May 19, 2026 | Drew Garty
Clinical development leaders have long been promised technology that could automate inefficient processes and eliminate workflow waste. While AI is the latest centerpiece of these expectations, true innovation is only as powerful as the value it delivers at scale.
As an industry we have not succeeded at true lasting change. Concepts like eSource and risk-based monitoring are not new to clinical research; many of these visions have existed for nearly 20 years and yet have not moved us towards our goal of more efficient and less costly clinical trials.
With operational pressures mounting, the focus must now shift from hype to delivery of innovation. By solving the most critical problems first with near-term realistic solutions, we can translate long-held visions into the scalable, standard practices that clinical trials actually need.
Innovating the whole ecosystem
While biopharma has proven that individual innovations can work in isolation, true transformation requires a connected ecosystem that functions across all stakeholders: sponsors, CROs, sites, and patients, and in compliance with regulatory authorities. We need to innovate in a way that allows for progress in one area to support the others – and we’re at a unique moment in time where technology and industry necessity have finally converged to make this possible.
Veeva continues to simplify and standardize clinical data while connecting stakeholders. Here is our vision to deliver value for sponsors, sites, and patients this year.
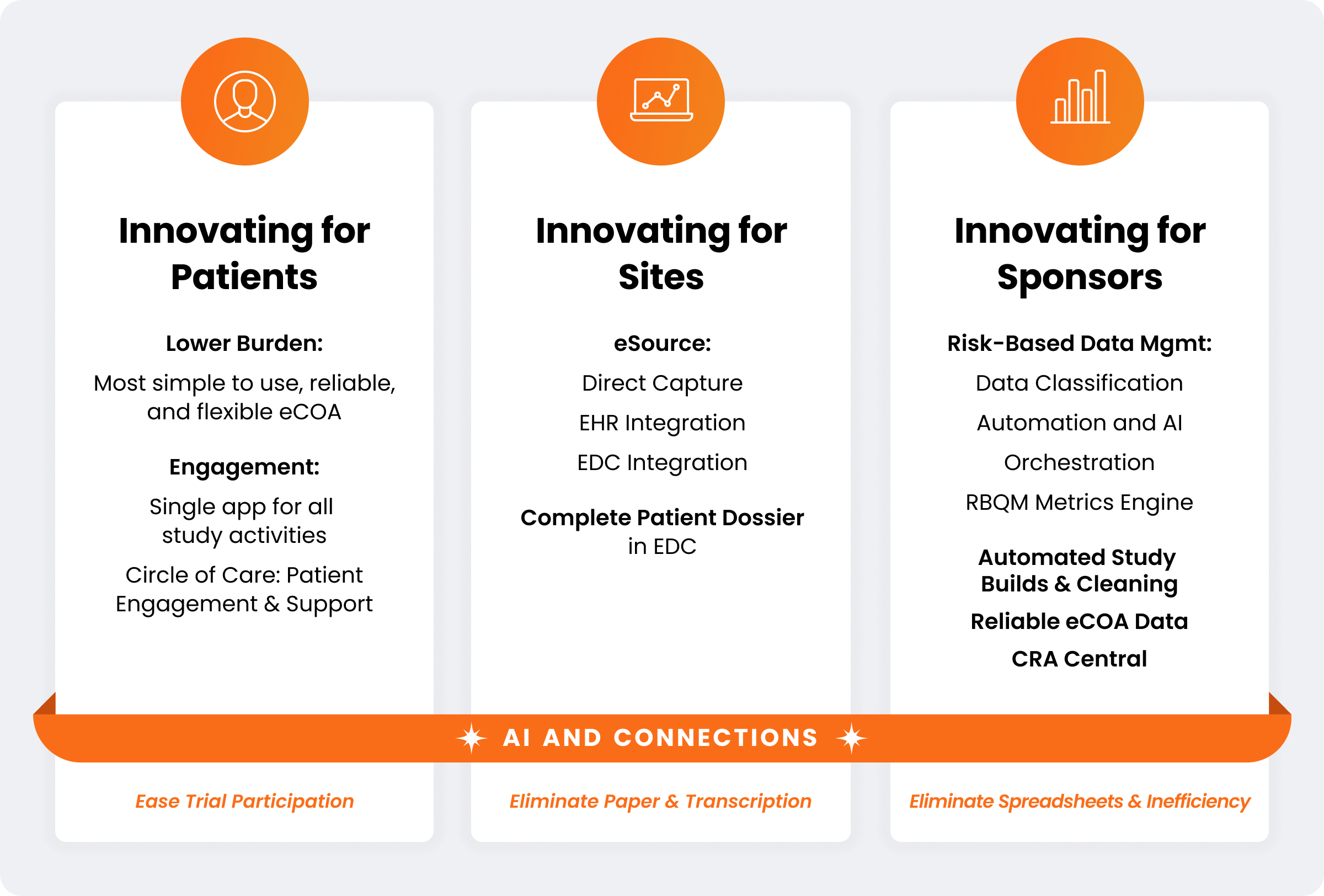
Innovating for sponsors and CROs: RBQM and RBDM
Industry research shows that data managers struggle with uniform cleaning across disconnected systems and workflows, spending too much time on inefficient manual tasks like data reconciliation, review, and cleaning. Data quality is at risk if inefficiencies aren’t addressed. Data management needs embedded automation and a commitment to risk-based initiatives.
According to ICH E6(R3) guidelines, risk-based quality management (RBQM) is governed by two principles: Quality-by-Design (QbD) and proportionality. QbD relates to the planning phase, and proportionality to the execution phase of a clinical trial. Risk-based data management (RBDM) is the application of these two RBQM pillars to the clinical data lifecycle.
RBQM should be employed to integrate clinical data with operational metrics, such as CTMS data, to orchestrate actions across functions. Veeva CDB will align with ICH E6(R3) by automating according to three data catalog dimensions: value, risk, and burden. It will be the accounting record of all the data being processed, including what it is, why it’s valuable, and what we need to do to protect it.
To automate any process with precision, we need a record of which data point supports which endpoints. For example, when a primary endpoint collected in an EDC system is identified as critical, it usually undergoes source data verification (SDV). Automating review and SDV plans tells data managers what to focus on, and eliminates the need for SDV altogether. Having a workbench such as Veeva CDB to deliver key risk indicator (KRI) and quality tolerance limit (QTL) metrics drives RBQM and RBDM, by triggering signals for downstream systems.
This will enable clinical development teams to scale RBQM and RBDM with precision.
Innovating for sites: eSource
Manual data entry from paper to EDC systems is still common practice at sites. eSource holds promise to resolve associated challenges with paper-based processes, including: duplicative entry and errors, the need for lengthy SDV, delayed data, and considerable burden on site staff.
Historical attempts at EHR-EDC integration have resulted in one-off builds that are costly and lack repeatable connections – a considerable time and effort sink for sites. eSource as a solution is not new, but has so far failed to scale. To succeed, we need to empower sites to carry out digital data capture at the point of patient contact, with their own systems and applications.
Veeva eSource will capture data once at the site where it flows directly into the sponsor’s environment, eliminating the need for transcription and associated verification. Sites will enter the three main data sources: EHR with FHIR mappings, direct data capture (through Veeva SiteVault), and, in time, document scanning using AI. Direct data capture will allow site staff to use the sponsor CRF for their site-specific data entry, and a FHIR-based approach enables precise, automated transfer of audit-ready data elements. This has a significant impact on eSource’s scalability.
EDC integration reduces the burden, cycle time, and cost of data collection by connecting directly with the sponsor system. Sites can take the initial protocol definition from EDC and augment it, without having to define the casebook from scratch. EHR integration means existing patient information can flow into eSource, so the site only needs to capture data that’s unique to the protocol. This all flows back to the EDC in real-time. Sites with existing eSource can work with their preferred tools and still connect to Veeva EDC, through an open API framework.
The industry has had proof of these concepts for years. Why will we now succeed with a scalable eSource? Veeva has the required specific infrastructure – SiteVault – and a wider connected ecosystem that brings together eISF, eConsent, CTMS, with the native interoperability between EHR and EDC.
Innovating for patients: Engagement and circle of care
Sponsors and sites both want to give patients the best study experience possible. But the complexity of trials, unreliable eCOA systems, and manual processes create considerable burden for patients. It’s important that we ease trial participation to improve engagement.
Veeva is focusing on not only giving patients more information, but easy access to timely and helpful information. We are expanding MyVeeva for patients, building on the existing framework to extend beyond eCOA, adding study task view, assessment instructions, support material, site communications, and training. It’s a single point of information for all clinical trial activities with the vision to increase patient engagement.
The framework for delivering value at scale
Historically, concepts like eSource and RBQM have failed to embed and scale because the industry lacked foundational standards and focused on only one stakeholder at a time. According to a Gartner survey, nearly two thirds of organizations are either unsure, or do not have, the proper data management practices required for AI projects. Too often, the promise of a new flashy tool distracts from building or fixing the data foundation.
Think of deep-space travel. Before you can even think about your destination or autonomous navigation, you need a stable launchpad, a flawless fuel delivery system, and expert ground control staff. If your “ground data” is shaky or unorganized it doesn’t matter how advanced your engine is. Building the foundation isn’t boring prep work, it is crucial to the mission.
“If you abandon these big idea rollouts after two years because the money has dried up or the project is ‘done’, you don’t see it systematically embedded in the organization. I think at the end of the day that’s a disservice to our patients.” Leianne Ebert Head of Global Data Management, Alcon
We’ve built from the bottom up, tackling the most critical problems and building a solid data foundation first, so that IT teams can deliver reliable solutions repeatedly.
The change management challenge
Humans fear change. Over the years, vendors have developed technology to solve industry problems, but when clinical development teams don’t use the tech to its full potential, then people become the bottleneck.
There will always be new technology. AI will follow the innovation adoption curve along its course and something else will emerge for the early adopters to become excited about.
Crucially, change management must be initiated in parallel with the development of any new technology to ensure the industry can actually adapt to these new ways of working.
“I think we live in pilots entirely too long. We need to actually enforce the rollout systemically across the organization. We have a responsibility as leaders to come back and show that the objective measures of success have been achieved.” Leianne Ebert Head of Global Data Management, Alcon
The time is now
Success requires a functional tech ecosystem that is fully adopted by sponsors and CROs, sites, and patients. Veeva’s unique advantage lies in having dedicated, separate teams for all three, which allows for a “triangulation” of expertise and support. By partnering with technology pioneers, we can take proven concepts and deliver them to the industry as mainstream standards, ensuring a controlled delivery of meaningful value.
Veeva can enable the industry with technology, and help organizations transition over time. Scalable innovation is a series of steps, rather than a single momentous leap. These calculated steps give change management the chance to keep up, but leaders must be prepared to lean into a more stable, connected future of clinical data.
Connect with industry leaders and explore how to put these strategies into practice at Veeva R&D and Quality Summit in Copenhagen or Boston.
