White Paper
Accelerate Lead Times Through Seamless External Collaboration in Quality
"Unified quality and using one Veeva platform has resulted in a better and more efficient collaboration with our customers.”
This white paper outlines:
- Why leading CDMOs are re-thinking how they collaborate with sponsors
- How sponsors are bringing partners into their systems
- External collaboration features within Veeva Quality Cloud that reduce lead times and benefit CDMO-sponsor partnerships
The rapid growth of the contract development and manufacturing organization (CDMO) market is underpinned by close partnership with biopharma companies for a wide range of services. But despite working side by side, collaboration between sponsors and CDMOs is not as efficient as it could be.
CDMOs often support the entire drug development process, from research to commercialization. Separate functions on both the CDMO and the client side, including quality, supply chain, manufacturing, and regulatory follow different processes, use distinct systems, and communicate through email, calls, and fax. Quality events and audits are often managed in disconnected environments, increasing compliance risk and potentially delaying product shipment decisions.
The reliance on external suppliers and partners is set to increase. By improving collaboration during quality processes, CDMOs will better support biopharma companies, optimize the cost of development and manufacturing, and ultimately get medicines to market faster.
When the two-way street is blocked
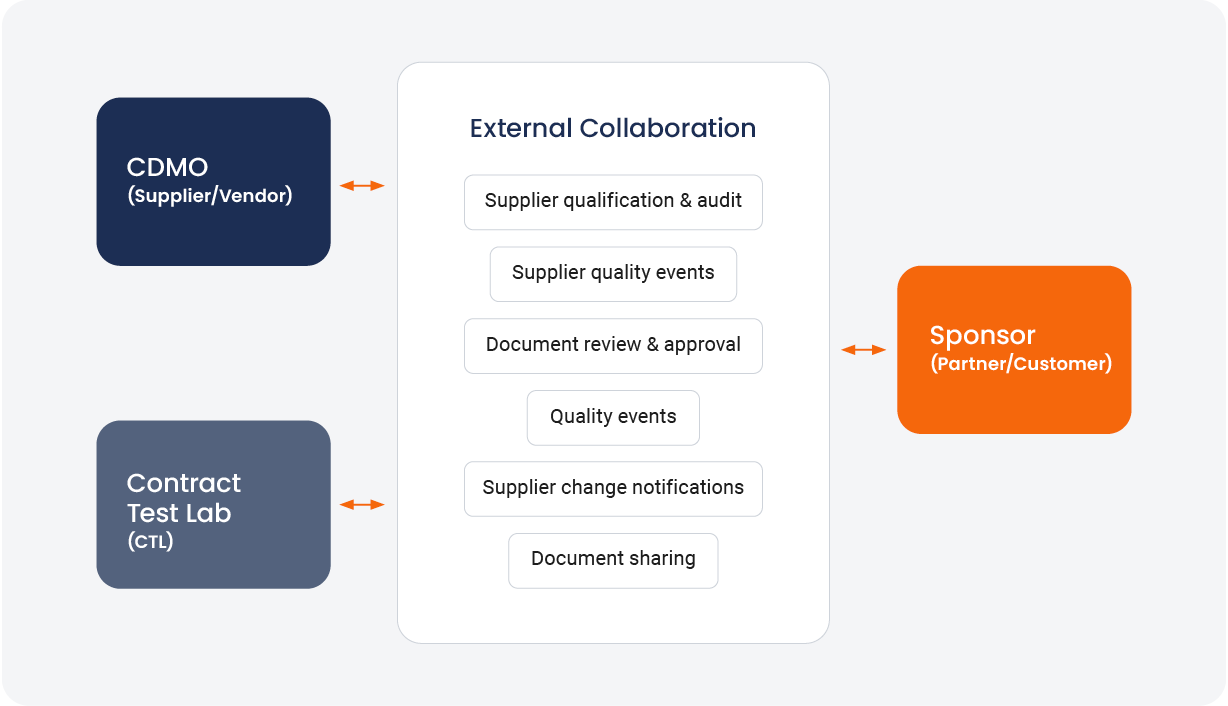
Several quality content and process activities require external collaboration [Figure 1]. Slow or inefficient collaboration can lead to disruption on the information highway, and issues with activities like quality agreements, closure of audit findings, and deviation management.
Poor collaboration also hinders service excellence and undermines trust. For sponsors, the outcome is a lack of visibility, compromised audit-readiness and efficiency, and a loss of control. For CDMOs, positive sponsor experiences are vital to becoming a supplier of choice.
FIGURE 1:
Network of collaboration for quality content and process activities
Success stories: Collaborating in a single system
CDMOs and pharma companies of all sizes benefit when they work in the same system that unifies applications, processes, and partners. Sponsors can bring suppliers and partners into quality workflows, collaborating seamlessly within their quality management system (QMS) and document management system. For CDMOs, contributing to quality activities directly in the sponsor system, or bringing the sponsor into their own system, adds significant value to the client relationship.
Here are some real examples of successful external collaboration.
Spotlight on sponsors
To improve oversight and operational efficiency, leading sponsors are moving away from manual communication by collaborating with CDMOs directly in their digital quality ecosystems. This fosters transparency, accountability, and a two-way partnership.
1. AstraZeneca
The top 20 biopharma works with 1,300 suppliers across manufacturing, testing, and distribution logistics. Quality processes were previously mostly manual, with email used to track progress and communicate. But a lack of visibility as to whether issues were being addressed lengthened lead times when closing quality events, such as deviations.
AstraZeneca decided to bring external partners directly into its systems, using Veeva QMS and Veeva QualityDocs to streamline external audits, document collaboration, and quality events. Now, investigation requests can be sent directly to suppliers for review and approval, with clearer accountability. “The suppliers manufacture as much product as a major site. So why wouldn’t we embed them into our system?” asks Andrew Sharrock, business IT system enterprise lead at AstraZeneca.
Automatically allocating licenses without the need for IT intervention ensures individuals can easily take action and complete tasks before their temporary access is removed.
"We can go into the system, put in a user who's a trusted partner, and then we can collaborate with them. I think it's a win-win because they're engaged with our system, it shows that we trust them and that we're embedding them into our processes.”
2. Neuraxpharm
With over 100 CDMOs collaborating on specialty drug manufacture, Neuraxpharm wanted to advance how it partnered with its network to improve compliance, inspection readiness, and oversight. The pharma company brought GxP documentation and training onto one platform with Veeva Training, while Veeva QualityDocs has streamlined content management and information sharing across all sites.
"With Veeva Quality Cloud, we can advance quality document management and compliance, including how we work with CDMOs.”
The CDMO perspective
After adopting a unified quality platform, CDMOs are transitioning from transactional relationships to strategic partnerships. As well as efficiency gains, these tools give CDMOs a business advantage.
1. Richter BioLogics
Using Veeva QMS, Richter BioLogics has streamlined previously manual processes (such as deviations), improving audit readiness and avoiding unnecessary delays during manufacturing. Knowing the real-time status on quality events makes it easier to address customer requests in pre-defined reports. Having access to simplified reporting also streamlines batch release preparation and boosts overall compliance.
"Some potential customers at one of our sites asked us about our quality system. After they heard we use Veeva Quality Cloud, they said ‘Okay, we don’t need to talk about the quality stuff anymore, we can talk about the manufacturing’. This is a real benefit for CDMOs.”
Richter BioLogics is planning to introduce external collaboration capabilities as the next phase of its quality transformation. Sponsors will gain direct access to the quality system, improving communication, customer service, and compliance throughout the manufacturing process.
2. Recipharm
The global CDMO relied on 12 different systems and paper-based processes to manage its quality and manufacturing operations across 14 sites. Most communication with clients and partners was via email and signature software services. Since using external collaboration features in Veeva Quality Cloud, Recipharm’s clients can review and approve records directly in the system, shortening approval timelines significantly. The CDMO also overhauled its change control process using Veeva QMS, reducing the number of review and approval steps from 14 to three.
"Unified quality and using one Veeva platform has resulted in a better and more efficient collaboration with our customers.”
3. Forge Biologics
From private biotechs to global NGOs, Forge is dedicated to working with clients on rare disease therapies. To reduce turnaround times, the CDMO needed to improve external collaboration during batch manufacturing, disposition, and client acceptance. If clients could start reviewing dispositions once manufacturing and quality reviews were complete, they could provide real-time feedback and approve product-impacting quality events.
Forge decided to unify quality processes and systems, and implemented Veeva QualityDocs, Veeva QMS, and Veeva Training in a connected quality management platform. By eliminating non-secure communication methods like email and shared drives, it can more easily involve clients in change control while ensuring compliant collaboration. For example, if the team comes across a quality event, it can add clients and share information for quick and easy review.
"External collaboration is a critical part of any CDMO’s business. At Forge, we’re really leveraging Vault to facilitate that collaboration with our clients, enabling faster turnaround times so patients can access treatments.”
4. Almac Clinical Services
Almac sought to consolidate multiple systems and connect its quality processes across people and partners. A unified quality strategy helped improve the company’s external collaboration with clients. Key selection drivers for Veeva QMS included real-time visibility into quality data, easier reporting and analysis of quality events, and reduced manual effort.
"Veeva QMS enabled us to standardize and streamline our processes internally, while also seamlessly extending them to our suppliers and customers.”
As Almac uses the external collaboration feature within Veeva QMS, it can invite clients and suppliers into the system to participate in specific tasks. The implementation team specifies which information external collaborators can view, based on needs and business priorities.
5. FUJIFILM Biotechnologies
Following a period of rapid growth, FUJIFILM Biotechnologies' goal was to move from traditional, transactional sponsor-CDMO relationships to true partnership. “We wanted to use a unified platform to build trust and better communication between sites and sponsors,” said Bradford Booth, director of global quality systems.
The Veeva external collaboration feature will allow FUJIFILM to share data, documents, and content with clients. This creates a better experience for sponsors, including those that have products at multiple sites.
"By leveraging a single platform across our network, we’ve moved from 'islands of data' to a seamless ecosystem where we no longer spend time searching for documents or clarifying details over email. This harmonization across sites doesn't just improve efficiency; it provides a 'superpower' of transparency that allows our teams to share data and metrics that mean the same thing to every site, ultimately accelerating how we serve our partners.”
How external collaboration works in Veeva Quality Cloud
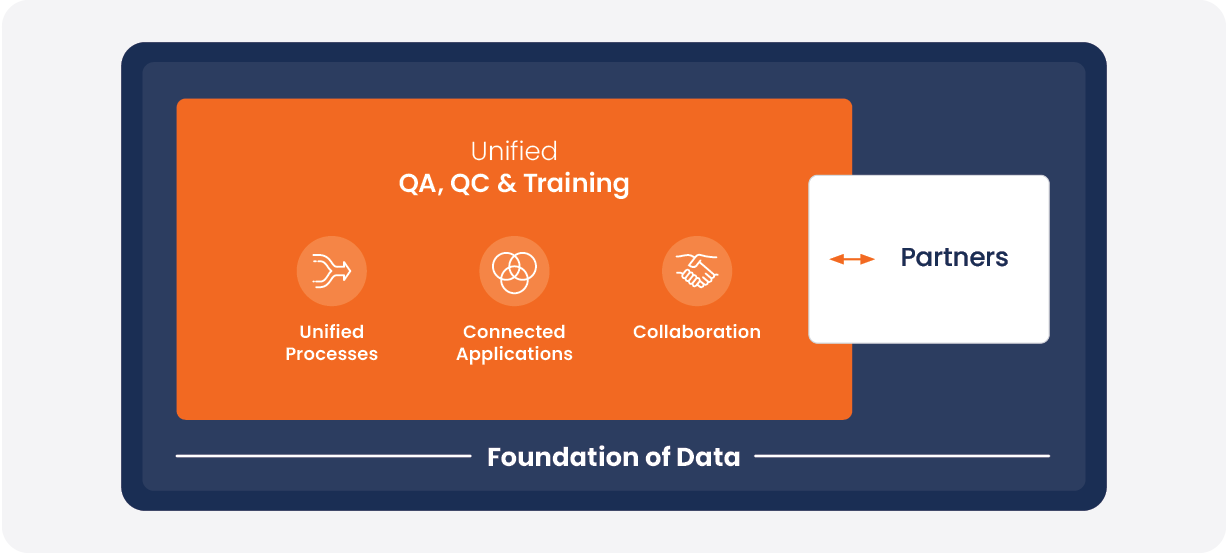
Veeva Quality Cloud consists of dedicated solutions for quality processes, content, training, and QC labs [Figure 2]. Together, these applications allow external partners to standardize their activities as follows:
- Veeva QMS:
Suppliers can respond directly to audit findings, SCARS (jointly investigate and prevent recurrences), supplier change notifications, and quality events (e.g., initiate investigations) - Veeva QualityDocs:
Internal and external parties can share information, such as procedures, policies, work instructions, quality agreements and certificates, and batch-related documentation, in a controlled manner directly within the system
FIGURE 2:
Veeva Quality Cloud supports sponsor-partner collaboration
Key collaboration use cases
Let’s explore some real-world use cases for Veeva’s external collaboration feature:
- External audits:
When CDMOs are manufacturing on behalf of sponsors, they need full audit trail readiness. But external audits are still heavily manual and a friction point for both parties.
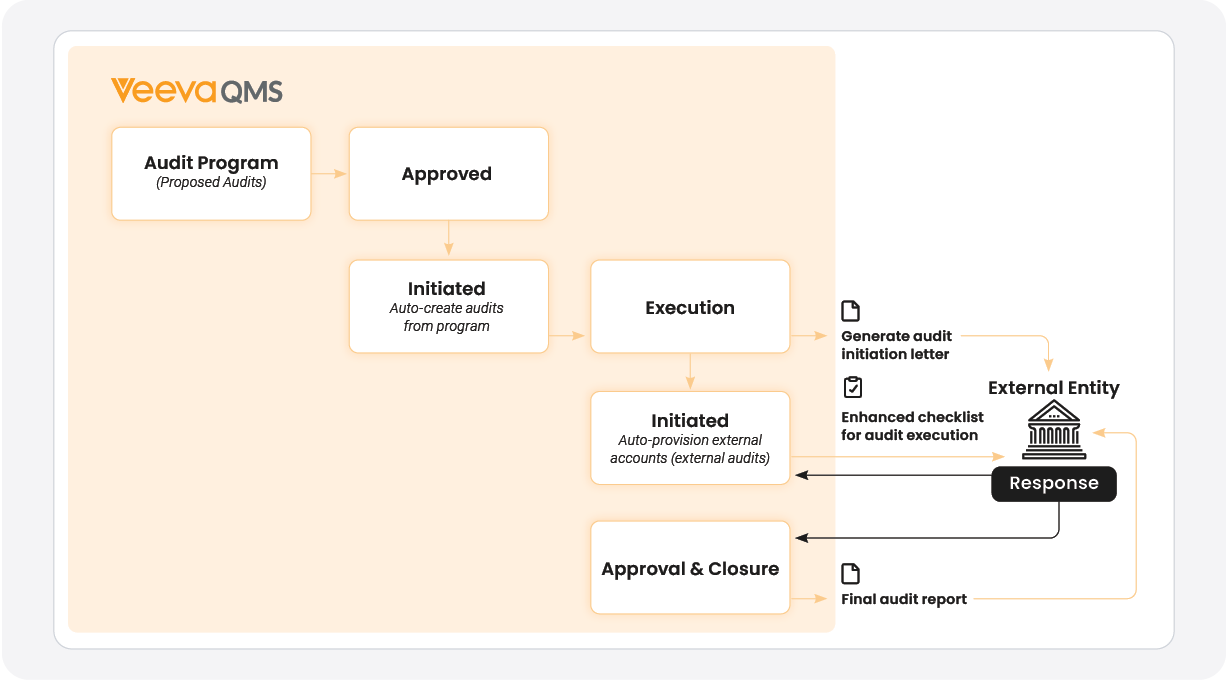
Sponsors can plan and schedule audits and document findings all within Veeva QMS [Figure 3]. To reduce the IT admin task of managing access for individual auditors and auditees, external audit reports are issued to third parties with temporary access. Through a self-provisioning wizard, auditees can respond to audit findings electronically, with access revoked after the task is complete or the finding answered. This eliminates the need for PDFs and spreadsheets, making the process more efficient for both CDMOs and their partners.
FIGURE 3:
Audit management workflow within Veeva QMS
- Deviation management:
CDMOs that want to scale quality operations must assess and improve their current processes. - Quality agreements:
Usually, sponsors and CDMOs send documents back and forth via email before reaching a final quality agreement, leading to poor traceability and the need for manual follow ups. With the external collaboration feature, the sponsor can draft, review, and approve documents such as quality assurance agreements with suppliers directly within Veeva QualityDocs. This eliminates the need for paper and email, and enables 21 CFR Part 11 compliance. - Automated notifications:
External collaboration tasks, such as reviewing and signing documents, investigation completion for a quality event, or answering a finding, experience fewer delays thanks to automated notifications. Direct notifications replace manual reminders, making it easier for sponsors to reply to suppliers and record their responses in a timely manner.
From transactional services to strategic partnership
The traditional ‘transactional’ relationship is no longer enough for leading CDMOs to maintain their competitive edge. By moving away from the friction of manual communication and disconnected systems, Veeva Quality Cloud transforms critical handoffs between sponsors and partners into a fast and transparent workflow.
Whether you are a CDMO striving to become a supplier of choice or a sponsor seeking total visibility into your manufacturing network, here’s a checklist that will help you align your quality processes with your business goals [Figure 4].
FIGURE 4:
Next steps checklist for sponsors and CDMOs looking to level up their external collaboration
| CDMO | Sponsor |
|---|---|
|
|
|
|
|
|
|
|
|
|