Blog
Changing Clinical Operating Models: Lessons from Large Biopharmas
Jun 16, 2025 | Angela Faircloth and Lauren Garson
Jun 16, 2025 | Angela Faircloth and Lauren Garson
Clinical operations leaders are assessing their operating models to determine how to increase data access and consistency across their portfolios.
This comes amid the industry’s multi-year move toward insourcing, as well as new guidelines such as ICH GCP E6(R3) that increase sponsor oversight expectations.
We recently met with clinical operations leaders of large biopharmas in the U.S. and Europe to discuss why and how they are bringing systems and processes in-house.
Assessing potential gaps in fully outsourced models
The majority of top 20 biopharmas have a primary operating model for clinical operations that relies on sponsor technology, including eTMF and CTMS, and sponsor SOPs. This ensures data access, enhances data quality, improves user experience, and speeds trial execution. Many large biopharmas outside the top 20 are also adopting sponsor-owned systems and processes.
The model below illustrates that using disparate systems across studies limits visibility, consistency, and data access. In this case, a sponsor would need to coordinate with five different CROs, potentially with varying data models, to get a view into their trial progress:
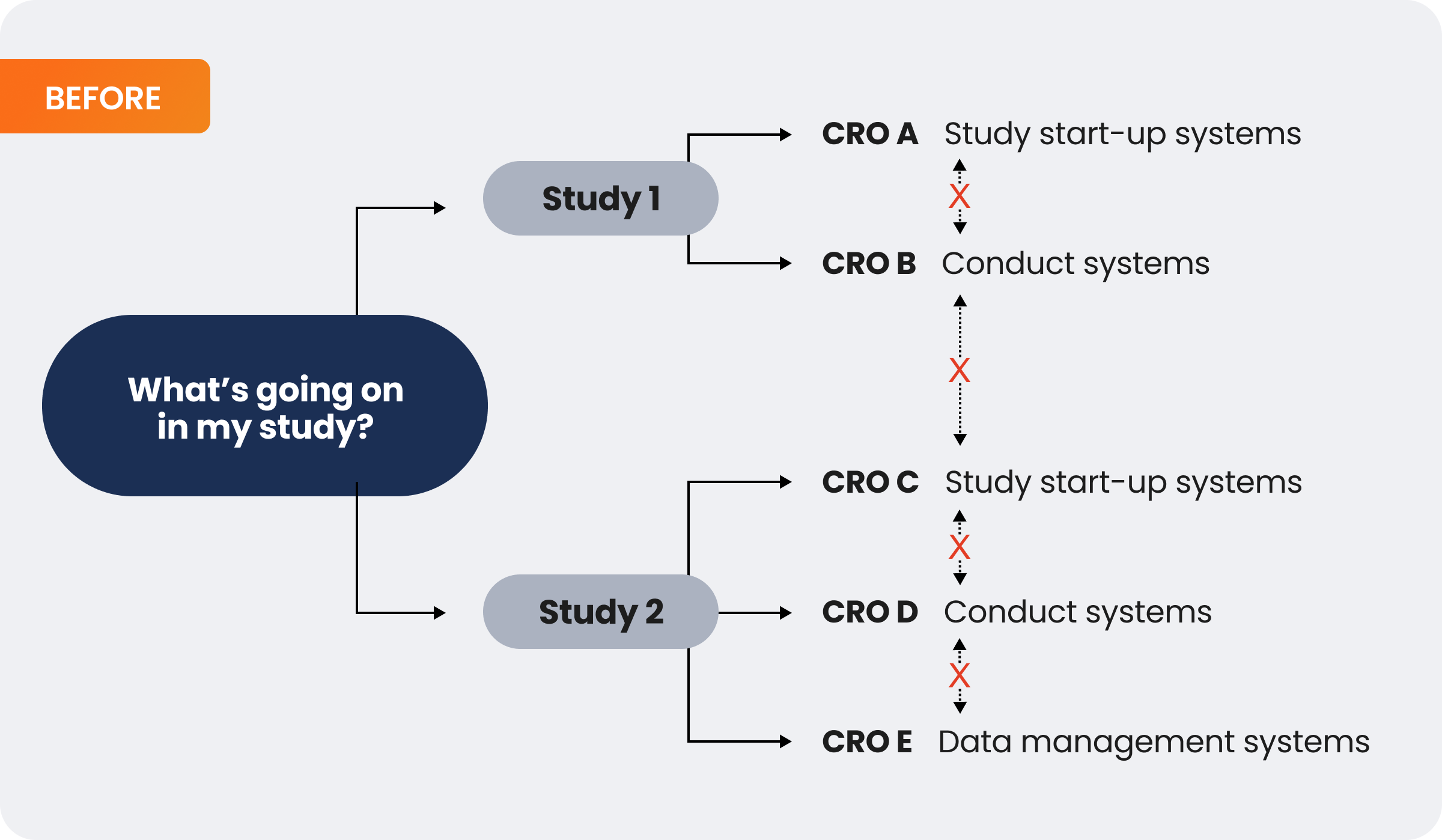
In contrast, having those same CROs work in a single, unified sponsor platform enables real-time oversight. In this case, a sponsor could access their data directly, and it would be standardized across systems and studies:
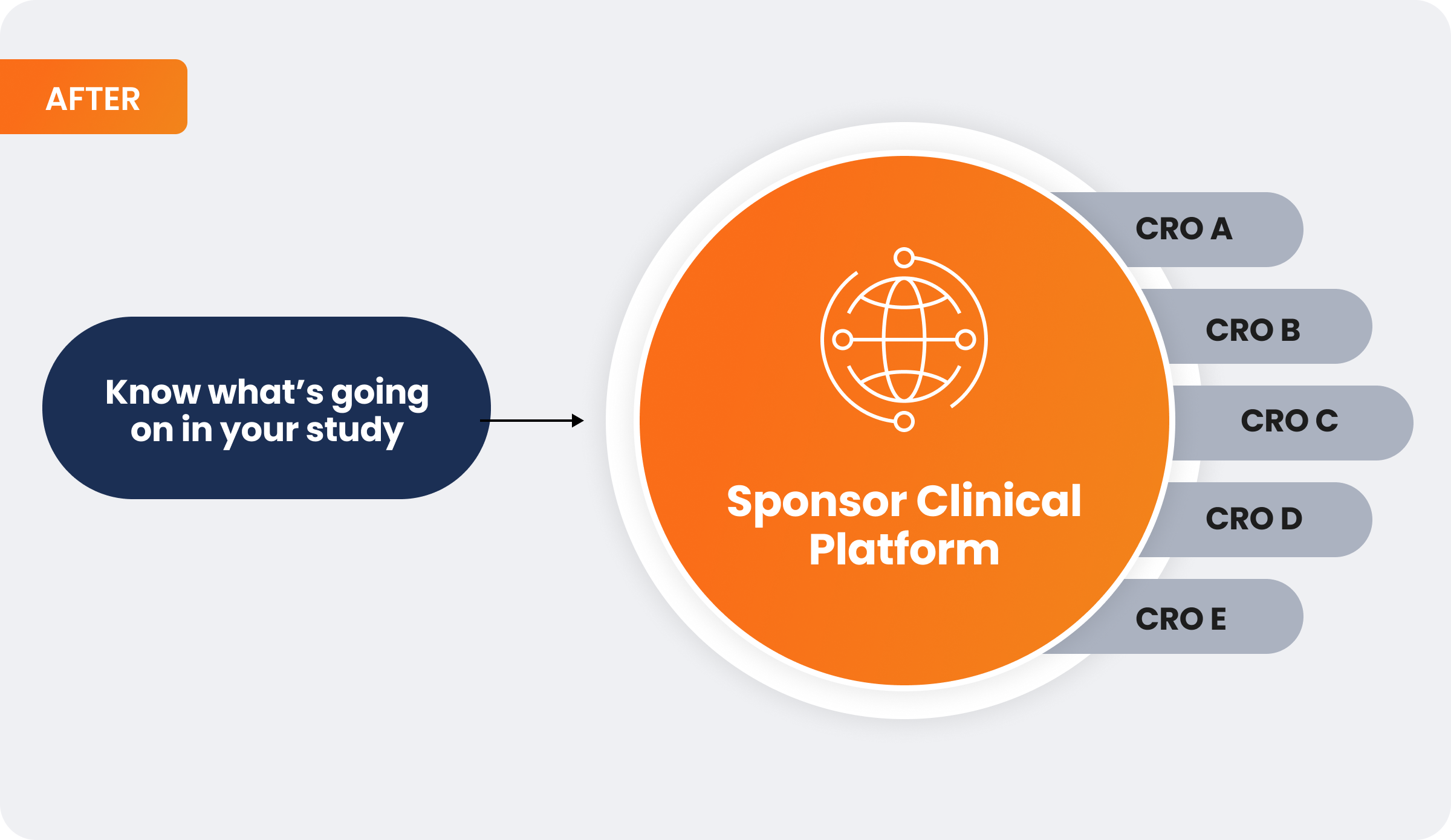
In a recent roundtable, most clinical operations leaders said they are moving away from fully outsourced (FSO) models where they don’t own most or any systems. They own key clinical operations systems to enable standardization across models, which can be decided on a study-by-study basis. Sponsors can use their technology for hybrid trials that have both insourced and outsourced functions and FSP studies with multiple CROs. That way, studies with multiple CROs are standardized in one set of systems. A sponsor can still use a CRO’s technology for FSO studies where all clinical trial activities are outsourced to a single CRO.
Implementing a strategic mix of insourcing and outsourcing
Sponsors may struggle to determine which systems to internalize, and when. Roundtable participants agreed that sponsors need a certain level of infrastructure in place to navigate the change successfully.
For one biopharma, internalizing medical monitoring introduced risk for outsourced activities because the rest of their operations were fully outsourced. Roundtable participants agreed that a minimum amount of infrastructure, including sponsor SOPs, are needed to bring activities in-house. This also includes a critical mass of clinical systems, such as eTMF, CTMS, and study start-up.
However, they also agreed that the benefits of insourcing outweigh the challenges, including the ability to standardize and access their operational data at any time. With standard data across systems, one biopharma can more easily centralize that data in a data lake for improved portfolio-level visibility. Another biopharma plans to leverage their data to inform decisions about site performance, selection, and asset modeling. Others plan to pursue synthetic data arms and data mining.
If you would like to discuss further lessons from large biopharmas and ways to successfully navigate a shift in your own operating model, reach out. Otherwise, watch this video to learn how one biopharma uses sponsor systems to standardize work across CROs, bring monitoring in-house, and establish a single source for study and personnel data.