White Paper
4 Ways Top Sponsors Build Strong and Sustainable Site Relationships
Discover how pharma leaders boost trials through site collaboration.
Over the past five years, there’s been an explosion of new technology introduced to improve clinical research site engagement. But these well intentioned solutions are often custom, standalone systems that require complex middleware and integrations. This forces sites to adopt dozens of sponsor-specific tools and build customized processes around them. This is just one of the reasons an estimated 60% of sites cease operations within their first year.
Although most sponsors understand the urgency and value of improving site relationships, it can be challenging to implement effective strategies that don’t exacerbate existing technology siloes. In this report, clinical operations and site engagement leaders at top 20 biopharma companies share how they’ve improved site collaboration despite these industry challenges.
Unpacking current conditions for clinical research sites
According to the Society for Clinical Research Sites, over 60% of sites use more than 20 systems daily. On average, site staff spend 5-15 hours per month learning how to use new technology. For busy research coordinators, this technology burden detracts from their top priorities: patient scheduling and being present during visits. Over-reliance on email and repetitive communication with sponsors and CROs limits the time site staff can spend with patients, often delaying start-up of vital studies.
A recent survey targeting over 10,000 research site professionals found that the top two technology challenges for sites are managing multiple logins and growing numbers of sponsor-selected vendors. One site commented “We have turned down several studies that use multiple technologies – both for subjects and ourselves. It just becomes too complicated.”
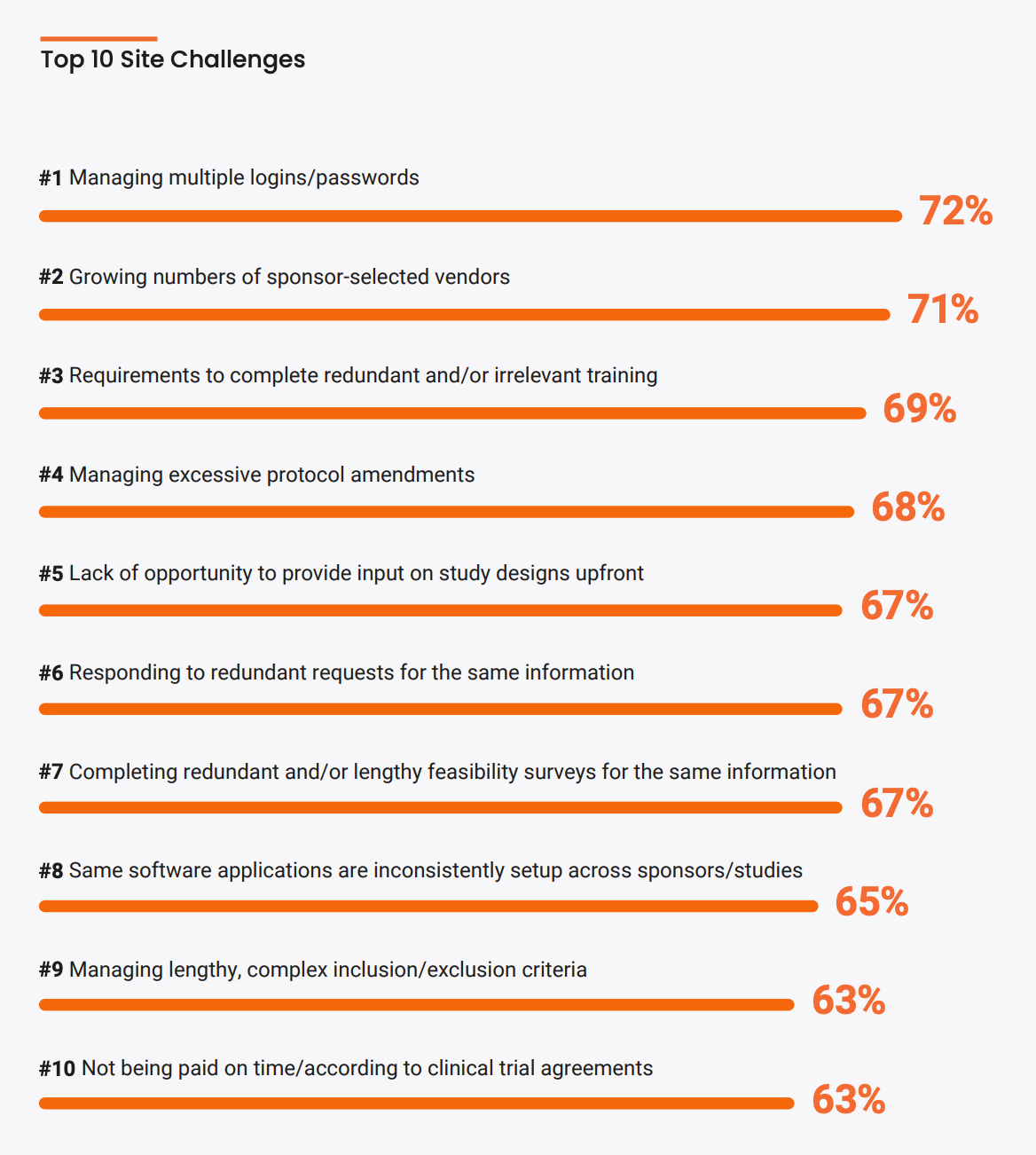
Despite tech complexities and dwindling site numbers, global sites are optimistic about clinical trials. A recent study from the Tufts Center for the Study of Drug Development found that site optimism is significantly related to the degree to which site staff feel that sponsors value and apply feedback from sites. This highlights the crux of the issue: an engaged site is one which has an open and collaborative relationship with its sponsors. Here, several leading sponsors demonstrate four key methods for improving site engagement:

Standardizing the site experience
Creating a consistent user experience is a top priority for most sponsors: they need a standard method for engaging with sites that allows flexibility toward the site’s preferences.
A senior director of global clinical solutions at one top 20 biopharma company is working to create an intuitive experience for sites. They explain how this approach could benefit the site employees and ultimately participants: “Sites need an easy system that they can navigate independently. They should be able to bulk upload documents and find the information required to support patients in real-time during the trial.”
The need for standardization is compounded by the number of different systems and passwords that sites must navigate daily. “We regularly hear from sites that each tool that sponsors provide requires knowledge and training,” they said. “Being able to provide a standard across sponsors removes some of the burden from sites and allows them to spend more time doing what matters most: helping participants.”
Another top 20 biopharma has a similar goal: become less sponsor-centric and give sites more choice when it comes to technology. The company’s head of clinical software development (COE) comments: "We think we are bringing the right solutions to sites, but it’s not just about our relationship with them. Sites are working with all of us.”
“It’s not just about our relationship with the site; they are working with all of us. That’s why we believe in Veeva, to get an industry standard for sites.”
Unified solutions that automate information flow across trial partners, processes, and systems are improving site collaboration without adding to the technology burden. These tools should have features like:
- Single sign-on to give sites easy access to all sponsor technologies through one ID
- A simple, free app to easily manage site content like ISF and delegation logs
- Streamlined information and data exchange to improve collaboration between sponsors and CROs
- Optimized training where sponsors assign tasks automatically before site visits and track on-site activities

Building collaborative relationships with clinical research sites
As an industry pioneer in site-centricity, Merck & Co., Inc., Rahway, NJ, USA (hereinafter "MSD") has worked to address the perception that sponsors have a top-down approach to their relationship with sites and do not consider their needs. Natalie Blake, senior director of MSD’s project management office at the global clinical trials organization (GCTO), explains the company’s approach: “To effectively partner with sites, we must give them a seat at the table and incorporate their valuable input into MSD’s decisions as a sponsor.”
“As we design and develop new technology, we want to incorporate site experience to ensure what we are designing is meeting end-user needs.”
MSD is moving away from process-driven site-sponsor relationships toward a consultative and dynamic mindset. To support this shift, the company implemented the Clinical Site Partnership (CSP) program, leveraging a network of 30+ global sites to develop clinical trial technology strategies that accommodate all study partners. CSP includes focus groups, live site observations, surveys, workshops, and user experience sessions.
Since implementing this program, MSD has garnered feedback on over a dozen initiatives in areas like clinical supplies and data management. The company also implemented a “menu” of operational enhancements to make it easier for sites to conduct clinical trials. CSP sites perform well with patient enrollment and comprise over 20% of MSD's oncology portfolio.
Sites participating in the initiative say that CSP opened lines of communication between MSD and monitors. Dr. Mustafa Erman, head of preventative oncology at the Hacattepe University Cancer Institute, describes his experience: “Being a part of CSP has allowed me to communicate freely and frequently with MSD to optimize processes. It’s also improved screening, recruitment, and patient care.”

Investing in site-centric technology
Part of building collaborative relationships with sites is providing technology that empowers success at every stage of the clinical trial lifecycle. One top 20 biopharma launched a joint effort between study start-up leaders and executive leadership to select KPIs that would provide better visibility and actionable insights to reduce cycle times. They identified metrics across 175 studies and created dashboards for each KPI to monitor progress by therapeutic area, country, and study. Using Veeva Site Connect and Veeva Study Startup together, the team was able to decrease site selected to site activated cycle time by 30% and decrease the time from when the start-up package is sent to activation by 36%.
The biopharma also implemented Veeva Site Connect to automate the distribution of safety letters, previously handled through over 10 million annual email notifications across 4,500 sites and 3,000 ethics committees. This has reduced manual steps in this process for staff by 70%.
A senior clinical director at the company emphasized that change management was key to the successful rollout, which included early stakeholder engagement, tailored training, and drop-in Q&A sessions. Beyond efficiency gains, the transformation redefined team roles, freeing staff from manual tasks and empowering them as country-level business administrators.
“Now that we’ve seen its success, we are working toward implementing this automated safety distribution process across our trial portfolio,” explains the senior director. “This will give every investigator a standardized experience regardless of study delivery model, and provide significant cost savings for us.”
Another top 20 biopharma has been on a similar site transformation journey. In just five months, the company implemented Site Connect, onboarded 8,400 global sites, and distributed 2.6 million documents.
“Site Connect does much more than simply exchanging documents and distributing safety data. It also enables us to share study announcements and contacts with sites, centralize links to all of the systems they need on a given study, and give them visibility into expected and completed payments.”
Since going live, the company reports a high adoption rate among sites, with many expressing a preference for participating in studies that use Veeva Site Connect. “We have significantly improved our team’s ability to track safety distribution. We know precisely who downloaded a document, and when they viewed it,” explains a company executive.

Better site training unlocks trial efficiency
To engage sites and streamline processes during study start-up and conduct, sponsors are looking toward optimizing site training. The global clinical capability strategy lead at one top 20 biopharma advocates for site simplicity and is focused on providing a user-friendly study training experience that meets the needs of individual sites and studies.
“I’m very mindful of the fact that site staff need to spend most of their time with patients,” she says. “As sponsors, we should ask ourselves: ‘How can we be more targeted in the training we deliver and our expected outcomes?’”
The strategy lead sought an automated, end-to-end training process in a validated system with these objectives in mind. The biopharma became an early adopter of Veeva Study Training, which automates and streamlines training for sponsors, CROs, and sites in one system. Their team implemented a multi-method approach to study training, offering pre-requisite reading of key study documentation, recorded modules, as well as targeted virtual and face-to-face interactions where there is additional study complexity.
The team has seen an almost 100% adoption rate among site users and provides the following tips to engage sites.

A connected environment for all
Making a strategic effort to refocus on the fundamentals is the key to improving site collaboration in the long term. Engaging sites and giving them a seat at the table will lessen the transactional nature of site-sponsor relationships and streamline study execution. The site-centric approach can be straightforward: prioritize site input and foster collaboration to ensure better clinical trial outcomes for all.
Veeva Clinical Operations provides an end-to-end environment and collaborative platform for sponsors, CROs, and sites. With integrated and automated workflows for start-up, training, execution, and close-out, clinical trials are accelerated with more efficient communication among cross-functional teams.
“That’s why we believe in Veeva,” concludes the top 20 biopharma’s head of clinical software development. “To get an industry standard for sites.”