eBook
Designing an Effective GxP Training
Environment for Life Sciences
A GxP training eBook for implementing an effective training program.
Foreword
This eBook includes a collection of articles providing guidelines for implementing an effective GxP training program.
In 2019, more than half of all GMP warning letters issued cited data-integrity deficiencies,1 many of which can be directly linked to inadequate GxP training programs. While improving training methods is surfacing as a priority in quality management, many life sciences companies are still using a paper-based approach to manage their training programs. Paper-based training may be effective for smaller, pre-commercial companies. However, as they grow and move towards a commercial state, compliance risks emerge as a paper-based approach fails to provide the audit tracking of training records. In some organizations, different functions adopt learning and delivery systems catered to their individual needs to solve the limitations of paper-training programs. This results in a fragmented training landscape without a single master location for SOPs, courses, and curricula. Absence of a single corporate-wide learning system hinders an organization’s ability to optimally deliver the right role-based qualifications to the right employees. The first article in this collection “Role-Based Training: Compliance and Qualification” discusses the limitations associated with legacy learning technologies, and how they prevent the effective delivery of specific role-based information to the right people at the right time. Read it here
Meeting strategic business objectives requires a modern training solution that centralizes training requirements across different functions and automates training delivery and assignments. By creating a single hub for learning, records, and knowledge, a modern training strategy can substantially increase training efficiency, effectiveness, and compliance. This helps lower both costs and data-integrity deficiencies, turning training into a key investment, and a strategic asset. “Make Training a Strategic Asset: Five Key Steps” outlines five essential steps in building the groundwork for a strategic training program, and how each step contributes to reducing friction in quality management. It explains how effective training programs can be an asset that directly influence quality metrics, instead of simply being an expense associated with risk avoidance. Read it here
Organizations that have adopted a modern training program are directly influencing their critical quality metrics, and enjoying a competitive advantage over other pharmas. “Improving Quality and GxP Training Effectiveness” shows the real-world impact for Xeris Pharmaceuticals, Dicerna Pharmaceuticals, and Foamix Pharmaceuticals in making training a strategic asset. By using a modern training system, all three companies were able to accelerate training development, streamline curriculum review and approval workflows, and execute faster internal audit preparation and onboarding of new team members. Read it here
ARTICLE 1
Role-Based Training: Compliance & Qualification
Corporate training professionals have been implementing role-based training concepts for decades, yet they continue to struggle with assigning the right amount of training content without incurring compliance risk. Modernized role-based training ensures the right content, reaches the right people, at the right time.
Learner roles do not always equate to a single job title, and should be based on a combination of job responsibility, function or other attributes such as job code or ID. However, delivery approaches using legacy learning technologies typically require learner roles to be the same as the job title. When 500 employees are defined by 500 unique learner roles, it is impossible to build a scalable training program that always delivers appropriate, contextual content. Trainers are forced to place individual learners into one or more groups – aligned within departments, business units or functions. With this model, it’s easy to over-train employees and partners while ensuring there are no compliance gaps. With modernized role-based training, learning objectives and corresponding training materials are aligned to the specific needs of an employee, partner or contractor.
For life sciences companies, role-based delivery is essential for efficient and effective training programs. It is important in commercial or leadership development training and mission critical for non-commercial areas such as quality, compliance, GxP and manufacturing. Limitations of legacy learning technologies prevent precise delivery of well-engineered content to specific learner roles, contributing to the following training-related challenges:
- Limited Visibility into Overdue or Incomplete Assignments: When learners don’t complete their training, they many not be qualified to do their jobs or capable of completing critical functions that is compliant with agency guidelines or regulations. Tracking and providing transparency on overdue or incomplete training allows all levels of the organization to gain visibility and mitigate risks.
- Lack of Content Mastery: Even with the perfect curriculum design – targeted to the right learner population, appropriately delivered and tracked – companies need to demonstrate content mastery. Adding assessments or observations around critical procedures can substantiate comprehensive knowledge and training.
- Training Gaps: There are several reasons that content-based gaps can occur – ranging from budget constraints to minimally qualified subject matter experts. Some training gaps have a compliance impact, others have functional repercussions. Easily identifying the right content to train the right roles eliminates compliance-related knowledge gaps.
- Over-Training: To avoid learning gaps, many organizations over-train their employees. With the limitations of many learning delivery platforms, it’s often easier to deliver more content to learners than manually configure focused curricula. Leveraging a training matrix that aligns learning objectives to roles, organizations can ensure learners consume, master and retain the right amount of information.
With a modern training solution, organizations can ensure the right content, reaches the right people, to do the right job, at the right time – even in a highly matrixed organization. To deliver maximum role-based compliance, consider the following capabilities in modern training solutions:
- Learner Role Visibility: It is essential that users are part of the right groups – learner roles and getting the right assignments. With visibility into learners and the groups users belong to, organizations can see that key operating procedures, policies and work instructions are reaching the right audiences. This ensures compliance in day-to-day job functions.
- Role-Based Automation: Automating assignments based on qualifications and job readiness helps learners stay up-to-date on training – even as they change roles, responsibilities or location. For example, if manufacturing plant employees change roles and need to operate a different piece of equipment, or a training administrator adds a new learner role to an employee, new training tasks should automatically be assigned. Technologies that leverage a role-based approach to training configuration can automate assignments based on change in attributes. Ensuring learners read critical operating procedures and there is traceability of job qualification status reduces compliance risk and improves quality outcomes.
- Unification: Allowing business critical applications to automatically trigger training tasks can accelerate quality processes and improve training outcomes. For example, corrective and preventive actions (CAPAs) frequently have a training impact. Identifying who should be trained is a manual process. Training materials that are organizaed by learner roles should be discoverable, flaggable, and associated with quality events or other business critical processes.
People are coping with information overload, and overtraining makes it difficult to focus only on content that will meet learning objectives. With appropriate role-based training, companies can automatically identify and deliver training based on a combination of attributes such as manufacturing site, department, and function.
Highly targeted training enables the right amount of information, delivered to the right person, at the right time, tom complete their job compliantly. As companies transition to modern learning frameworks, such as microlearning, it is critical to find learning technology that is flexible and support role-based training.
ARTICLE 2
Make Training a Strategic Asset: Five Key Steps
Simplified role-based training can lead to better quality metrics and compliance.
It is no secret that staff training remains a weak spot in many corporate strategies. In 2017, 30% of the 40 warning letters that FDA issued for data integrity deficiencies directly referred to inadequate training or training requirements.1
A number of challenges prevent life-sciences companies from developing more effective training programs. One major problem is the way training-related data have been traditionally organized. For example, most pharmaceutical and biopharmaceutical companies use one system to manage standard operating procedures (SOPs) and other knowledge-management documents, and a different system to manage training. Ideally, the two areas should be connected, or a barrier will be created between activities that should be closely aligned.
For example, consider the specialty pharmaceutical company Tolmar, which had built a custom application to manage and track compliance-based training requirements, but used a cloud-based system to administer various other types of training. The company’s in-house learning management system (LMS) required extensive configuration and integration, as well as ongoing validation support.
“We have around 1600 documents that 700 employees across the organization need to be trained on. Managing this workload across two applications—our cloud-based document management system and in-house LMS—is complex and requires a lot of overhead,” says Joe Miller, Tolmar’s vice president of information technology.
A similar lack of alignment is often seen between training systems and corporate business objectives, which can make it difficult to link training outcomes with concrete goals such as reducing manufacturing-related deviations or other quality events. Rather than viewing training as a corporate expense, managers should be able to see how effective training programs directly influence critical quality metrics. As a result, more life-sciences organizations are rethinking training, and starting to view it as a strategic part of quality assurance and control, and overall business goals.
"We have around 1600 documents that 700 employees across the organization need to be trained on. Managing this workload across two applications—our cloud-based document management system and in-house LMS—is complex and requires a lot of overhead." – Joe Miller, vice president of information technology, Tolmar
A growing number of pharmaceutical companies are now working to modernize training programs, to align training with compliance documentation and corporate strategy. This requires unifying processes across quality, content, and training systems for improved quality management. In a unified, end-to-end approach to training, users first identify and revise the documents that would be most significantly impacted by a deviation. Then, when a quality event does occur, the system automatically triggers and assigns training tasks to the right people. In this scenario, training is connected to document versions, change-control processes, and quality events, helping to support broad organizational goals to improve quality metrics.
Getting to an end-to-end approach will take some time and effort, but following the five steps below will lay the groundwork for making training a strategic asset.
Develop a bill of learning that attaches trainable behaviors to key quality metrics
Bridging the gap between a business goal and training starts with a bill of learning, which breaks the goal down into discrete, measurable learning objectives for a specific skill set. Educational initiatives can then be tied to a company’s strategic direction, helping improve critical metrics. In addition, a bill of learning can help demonstrate the impact that training has on the overall business.
In this bill of learning (Figure 1), a pharmaceutical company has encountered out-of-specifications (OOS) due to failed lab tests. The quality event is ruled to be not a lab issue, but a manufacturing deviation resulting from contaminated material introduced when equipment was not properly cleaned. This scenario has happened before, so in response, the company establishes a strategic goal to decrease deviations, specifically related to proper cleaning techniques.
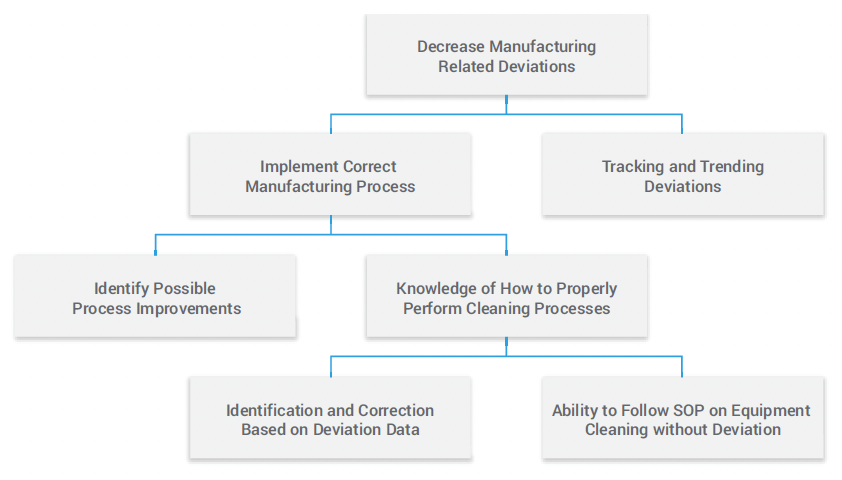
Figure courtesy of the author.
"With a bill of learning, companies can break a strategic objective into specific learning components and link the learning outcomes back to the organization." – Karl Kapp, director of the Institute for Interactive Technologies and professor of instructional technology at Bloomsburg University
The objective to decrease deviations is divided into the actions needed to meet this goal, such as implementing the correct cleaning procedures related to the manufacturing process. To implement the right manufacturing process, quality teams want to identify process improvements and increase employees’ knowledge of performing a process properly. The company decides that to reinforce knowledge of how to execute a process—in this case, cleaning techniques—individuals’ ability to follow SOPs without deviation must by improved.
This is where training comes in, to identify and correct quality professionals’ ability to follow the procedure. Ultimately, the bill of learning makes it easy to understand the impact of not following proper SOPs, which helps reinforce the importance of following approved processes without deviation.
“With a bill of learning, companies can break a strategic objective into specific learning components and link the learning outcomes back to the organization,” says Karl Kapp, director of the Institute for Interactive Technologies and professor of instructional technology at Bloomsburg University. “In doing this, companies effectively connect quality and compliance, gathering metrics that can be measured against corporate objectives to continuously improve quality processes.”
A modern solution unifies training and quality management, enabling teams to track quality metrics and link them back to training to ensure effectiveness. Nationally or globally dispersed organizations can bring together SOPs, quality processes, and training with complete transparency. In this example, companies identified documentation related to this specific deviation and built training around the recurring behavioral gaps. With a bill of learning, training is also attached to a corporate objective to ensure alignment across internal and external stakeholders.
“At Tolmar, one of our quality goals is ‘do it right the first time,’ and a cloud-based training solution provides a strong foundation for this goal,” says Miller. “Many pharmaceutical companies have a program or process for SOP training, but most of these programs are inefficient. A training system in the cloud that’s connected to quality processes and content helps streamline our training, so we are more confident that critical documents have been read and understood.”
Define roles for role-based training
Once a bill of learning has been established, the next step is to specify learner roles—the foundation for role-based training. Modern role-based training uses a combination of job responsibility, function, and hierarchical level within the department or organization.
With legacy technologies, learner roles are often exclusively tied to a specific job title or ID, limiting the ability to deliver precise content to each person. People are often undertrained or placed in more than one group and over-trained. Both situations present compliance risks. Without the ability to deliver appropriate, contextual content, it is almost impossible to build a flexible and scalable training program while ensuring compliance.
For example, without role-based training, every employee in a quality manufacturing department receives the same curriculum, such as training on 85 SOPs and work instructions. With flexible, role-based training, organizations deliver tailored content to specific roles such as a quality manager, quality assurance associate, or documentation specialist. Instead of being trained on all 85 SOPs and work instructions, a quality assurance associate would only train on the 25 that are specific to his or her role. Assigning a specific curriculum to each role helps to reinforce the learning objectives that directly connect to those responsibilities.
Defining learner roles is a critical first step in implementing a role-based training curriculum, and it starts by asking questions like, “Is each role specific to one department, job, or function, or combination of these attributes?” or “Which roles are applicable to the behaviors identified in a bill of learning?” The answers will help teams tailor training programs to ensure that the right content reaches the right people, to do the right job, at the right time.
Modern training solutions connect training to learner roles in an end-to-end process within the quality system. A quality document is tagged as required training, and, in the event of a deviation, the content is revised via change control. The revised document is then automatically reassigned to learners as a training task. For example, when the equipment cleaning process is modified, the SOP is flagged as required training. With role-based training, the SOP training task will automatically be sent only to the employees that use the equipment, instead of everyone on the manufacturing floor. In the end, this approach allows companies to deliver the right content to more precisely segmented audiences without over- or under-training, increasing efficiency and compliance.
“The ability to track a user’s training status or assessment for any training document enables management to ensure only those qualified to perform a particular operation can do so,” says Miller. “For example, if someone is out for the day, it’s easy to find other qualified employees to perform a task, helping minimize compliance risk and improve daily operations.”
Use microlearning to connect critical content to learner roles
After determining learner roles, organizations can apply microlearning techniques to break down larger and more complex SOPs to develop hyper-focused content for their role-based training programs. For example, if many deviations have been linked with a failure-to-follow a key SOP, companies can then divide that SOP into smaller, targeted learning assets to help staff focus on the necessary skills. An organization could create a short, two-minute video to demonstrate only a specific cleaning procedure to support a longer SOP. Since each task is connected to a larger instructional objective, microlearning reinforces the right behaviors for better performance that improve strategic quality objectives.
“Performance support and learning are inextricably linked,” says Kapp. “In any organization, we learn because we want a certain outcome. We want learners to perform an action correctly, so microlearning can really be an invaluable tool for supporting improved performance.”
Microlearning in a unified quality system helps make the learning journey holistic, instead of introducing learning as a series of one-off events. Once a quality procedure has been revised, the system automatically re-assigns training for that SOP based on the pre-determined learner roles. This approach helps ensure that learners are not only reading and understanding documents, but also applying that knowledge to their daily tasks effectively.
“We want to expand our program to go beyond simply training on SOPs and see if specific training has a real impact on an individual’s performance,” says Miller. “With our cloud-based application, we can use concrete evidence, such as results from on-the-job training, to assess an employee’s comprehension. Doing it right the first time will help us avoid rework and costly errors.”
Deploy training in the flow of work
Microlearning can only be as effective as when, where, and how it is deployed. Companies can expect better results from training programs by shifting from individual, content-driven events to learning that is deeply contextual, social, and embedded into real work.2
Considering how much information is consumed via technology every day, meeting learners where they learn best—in the flow of their work and day-to-day life—is crucial. The average person checks his or her smartphone nine times an hour and pays attention to specific content for less than seven seconds.2 In fact, smartphones dominate as learning technology, and recent research has shown that 70% use their mobile devices to learn.3
Where and when learning takes place should also be considered when deploying training materials. A large percentage of learning happens during the workday, with 27% of learners consuming content during the work commute and 42% at work. Since more than half of individuals learn at the point of need, microlearning can greatly impact learning objectives by delivering learning events more rapidly and frequently.
“When a learner needs to retrain on the appropriate steps to execute an action, they can access training in the flow of their work when they need it most, without delay or interruption,” says Kapp. “This is an example of how microlearning and the strategic goals of an organization can come together to create the right learning environment.”
One solution for training enables both learners and trainers to focus on the right content, at the right time, across devices. More pharmaceutical companies are using tablets in manufacturing facilities so people can access the relevant work instructions they need at any station on the shop floor. Employees ensure they are performing the right action while they work and can reference training content at any time, on any station. For example, a video that demonstrates cleaning procedures can be available via a smartphone so learners can train on how to clean machinery as needed. By connecting learners with training content at the time of need and according to their learning habits, companies can better change behaviors to decrease quality events.
"When a learner needs to retrain on the appropriate steps to execute an action, they can access training in the flow of their work when they need it most, without delay or interruption. This is an example of how microlearning and the strategic goals of an organization can come together to create the right learning environment." – Karl Kapp, director of the Institute for Interactive Technologies and professor of instructional technology at Bloomsburg University
Generate insight and take action to realize measurable impact
Many organizations have encountered difficulties generating comprehensive reports around training or qualification tasks and how they are related to compliance. Training tasks live in a different place than the training content, such as in a document management system, email, or in multiple learning platforms, reducing visibility.
End-to-end insight in a unified system enables companies to understand how training is related to quality objectives to make better, more informed decisions. Once a training initiative has been completed, teams generate reports that include which critical content and version are associated with deviations, when corresponding training materials linked to a deviation were consumed, and whether the number of deviations decreased as a result.
With quality processes, documentation, and training unified in one solution, quality teams can then effectively report on strategic metrics and the return on investment of the training program. A bill of learning in a unified system provides a direct correlation from the discovery of an issue to the deployment of training and a tangible process improvement.
“Direct integration between our training, documents, and processes allows us to measure the impact of training and determine if training is helping us meet strategic goals,” says Miller. “We can schedule training for new and revised documents, link quality events like corrective and preventive actions to training tasks, and measure the results. These new capabilities, only possible with a unified platform, will eliminate manual processes and allow us to stay dynamic and better align our processes with quality objectives.”
Strategic training to improve quality metrics
Corporate learning in life sciences has the potential, not only to improve productivity and reduce errors, but to also become an important source of strategic, competitive advantage.3 These five steps offer a starting place to drive continuous process improvement in quality. By continually breaking down a strategic objective into specific learning components, companies teach to those components and apply the outcomes to the organization. At the conclusion of the process, teams provide metrics that are directly associated with the strategic advantage, measuring improvement and building better quality programs around a specific learning objective.
The cloud is enabling the industry to bring training and quality processes together and transform quality management. The results of doing so include saving time by accelerating internal audit preparation and onboarding team members faster, as well as improving compliance as more quality professionals can execute their jobs better. Correlating training results to quality objectives will enable organizations to make more informed decisions and rethink training as a critical business strategy.
By connecting quality management and training in a single application and applying modern learning techniques, life-sciences companies can create an effective training program that’s measurable across the organization.
2 S. Penfold, “Seven Mobile Learning Design Strategies: Tips, Examples, and Demos,” elucidate.com, April 19, 2017.
3 J. Bersin, “How Corporate Learning Drives Competitive Advantage,” forbes.com, March 20, 2018.
ARTICLE 3
Improving Quality and GxP Training Effectiveness
At the 2019 Veeva R&D Summit, experts from Foamix, Dicerna, Xeris, and Xencor came together to discuss opportunities for improving quality and GxP training effectiveness.
Automation drives training compliance
When Xeris grew from 12 people to over 200, demonstrating training compliance using paper became difficult. “Auditors and inspectors needed evidence that our employees were appropriately trained to perform their jobs,” said Ashley Gregory, corporate training manager at Xeris. “Not knowing if employees met essential requirements and completed the training on time became a compliance risk. A stack of paper couldn’t give us any insight.”
Xeris gained that insight with Vault Training, an industry-specific learning management application. Ashley added, “Now we can distribute the training on time, follow up on training completion, and trace job qualification status.”
While industry-specific learning management solutions improve training outcomes, most life sciences companies use them in disparate environments. Having different systems for managing standard operating procedures (SOPs), knowledge-management documents, and training prevents companies from developing effective training programs.
Unified document management and training solution improves training effectiveness
Unifying industry-specific learning technology with other quality applications brings unique benefits. For example, automatic assignment of training based on quality events such as CAPA workflows or document changes improves efficiency and quality.
Anthony Davis, global head of quality and compliance at Dicerna, shared how configuring curriculum review and approval workflow in a single platform streamlines document changes, related curriculum updates, and subsequent training assignments.
"A unified training and document management solution automatically assigns retraining based on continuous updates of controlled documents, increasing visibility and training efficiency." – Maggie Enterline, senior manager at Foamix
“In multi-system environments, training administrators have to manually track document version changes and determine when retraining is needed,” said Anthony. “A unified environment manages document and curriculum lifecycles in the same system, accelerating and simplifying training development, administration, and delivery.”
Foamix described how using Vault Training with Vault QualityDocs strengthened quality management across their US and Israeli sites. According to Maggie Enterline, senior manager at Foamix, “A unified training and document management solution automatically assigns retraining based on continuous updates of controlled documents, increasing visibility and training efficiency.”
Unifying processes across quality, content management, and learning management ensures that training remains aligned with compliance requirements and corporate strategy. The results are faster internal audit preparation and onboarding of new team members, as well as better training and quality outcomes.
Author
Kent MalmrosSr. Director of Vault Training, Veeva Systems
Kent has spent the majority of his career delivering technology-enabled training solutions to life sciences, holding leadership positions at industry leading companies such as AdMed, ClearPoint (Red Nucleus), UL EduNeering (UL), and now at Veeva Systems. As Senior Director, Kent is responsible for the product strategy, market facing operations, and customer success of Veeva Vault Training.
For more information about a unified training environment, visit our website veeva.com/vault-training/.