Veeva OpenData Ohio TDDD Compliance
Simplify Compliance
with Ohio TDDD
Streamline compliance and deliver
samples without delay.
Learn how Alnylam Reached a Broader HCP Audience
Veeva OpenData Ohio TDDD Compliance
Veeva OpenData streamlines compliance with Ohio Terminal Distributor of Dangerous Drugs (TDDD) requirements so you can stay focused on commercial execution.
Benefits
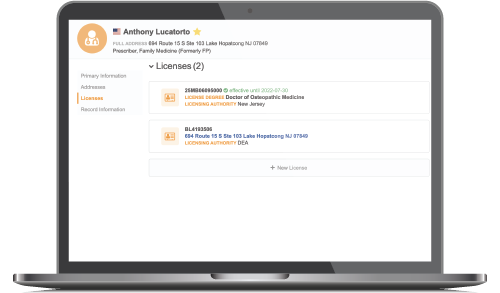
Reduce Compliance Burden
Use built-in compliance workflows and automated validation checks to allow field teams to detail prescribers and sample products without delay.
Quickly Adapt to New Regulations
Access verified cross-referenced license data and stay current with changing regulations as they take effect.
Stay Focused on Your Business
Combine up-to-date licensure information and affiliations data to ensure compliance and keep reps focused on commercial execution.
Resources for Ohio TDDD Compliance

Press Release
Veeva OpenData Helps Meet Compliance Requirements in Ohio

Video
Ohio TDDD License Verification Requirements

Webinar
Ensuring Compliant Sales Engagement